The influence of accommodation on choroidal thickness: a pilot study
Purpose: The aim of this study is to characterise the changes in choroidal thickness during and after a prolonged accommodation period using optical coherence tomography (OCT).
Subjects and Methods: This study used 24 test subjects (age 28.2 ± 2.6 years), with n = 18 assigned to the moderate myopic group (MSE: -2.6 ± 1.76 D) and n = 6 to the emmetropic group (MSE: -0.07 ± 0.24 D). The test subjects underwent an accommodation relaxation phase (ten minutes), an accommodation phase (twenty minutes of continuous accommodation), and another relaxation phase (ten minutes). Seven OCT scans (Topcon Corporation, Tokyo, Japan) were taken along a horizontal axis on each eye at five-minute intervals. For each tomogram, the subfoveal, temporal and nasal choroidal thickness was determined manually at three measurement points (2 mm apart) using a measurement tool. To trigger the process of accommodation, a mobile phone was attached to the side of the OCT device and the display was projected into the measurement beam path of the OCT via a semi-transparent mirror (demand of accommodation = 5.0 D).
Results: During accommodation, the choroidal thickness decreased significantly by an average of -8.06 ± 7.33 µm in the subfoveal region, -4.00 ± 11.00 µm in the nasal region, and -5.29 ± 12.52 µm in the temporal region (each with p < 0.01). Furthermore, emmetropic eyes showed a significantly stronger decrease in the subfoveal region (-13.08 ± 8.58 µm) than myopic eyes (-6.39 ± 6.06 µm). In the ten minutes following the accommodation phase, the choroid returned to its initial thickness, showing no significant difference (p = 0.80) in the degree of recovery between myopic and emmetropic eyes.
Conclusion: The study shows that the process of accommodation is associated with a decrease in choroidal thickness. The results further suggest that accommodation in myopic eyes entails a smaller decrease in choroidal thickness than in emmetropic eyes. The apparatus design developed specifically for this study to project a smartphone display into the measurement beam path of the OCT device is suitable for stimulating accommodation over a longer period while performing repeated OCT scans.
Introduction
The global increase in myopia and the associated ocular conditions pose a major challenge.1 According to estimates, almost 50 % of the world‘s population will be affected by moderate myopia and a further 10 % by high myopia by 2050.2 Disproportionate eye growth is generally the most important cause of myopia, although its triggers have not yet been fully researched. It is recognised that eye growth is controlled by genetic factors and visual input3 and that near work favours the development of myopia.4 However, it is less known whether the choroid, as a structure of the posterior eye segment, has an influence on the development of myopia. This assumption is reasonable because, on the one hand, the sclera is not innervated and, on the other hand, eye growth is initiated locally in the area of the posterior eye segment.5 The choroid seems to be involved in the regulation of eye growth as a secretory tissue,6,7 whereby a connection between eye growth and a change in choroidal thickness could be demonstrated in pharmacological experiments and via visual manipulation. Accelerated eye growth is associated with a (spontaneous) decrease in choroidal thickness, whereas, on the other hand, a slower eye growth is connected with an increase.5 Complex biochemical processes and the choroid, which is difficult to access in the living eye, make it difficult to carry out appropriate studies and to draw concrete conclusions from the results. However, various authors have been able to show that a blurred retinal image can lead to a change in the thickness of the choroid. This suggests that there is a connection between the process of accommodation and changes in the choroid. So far, however, only a few studies worldwide have shown the extent to which the process of accommodation affects the choroid mechanically.8-10 The aim of the present study is therefore to characterise the changes in choroidal thickness during and after continuous accommodation. A comparison between emmetropic and myopic subjects should also provide information about the possible connection between the choroid processes and myopia.
Study subjects and methods
This study examined the change in choroidal thickness during the process of continuous accommodation. For this purpose, the choroidal thickness of several test subjects was measured repeatedly using optical coherence tomography (OCT) while the subjects were exposed to an accommodation stimulus over a defined period of time via a specially developed mirror apparatus. 24 test subjects (11 male, 13 female, age 28.2 ± 2.6 years) were included in the study. Using a randomised selection, we measured 10 left eyes and 14 right eyes. According to the WHO classification of ametropia,11 18 eyes were assigned to the moderately myopic group (MSE: -2.6 ± 1.76 D) and 6 eyes to the emmetropic group (-0.07 ± 0.24 D). The inclusion criteria were a best corrected visual acuity VCC ≥ 1.0 (decimal) and an accommodation amplitude ΔAmax > 6 D. The exclusion criteria included all types of ocular or general diseases that could have an influence on vision or the ability to accommodate, uncorrected astigmatism > 0.75 D and having undergone refractive surgery.
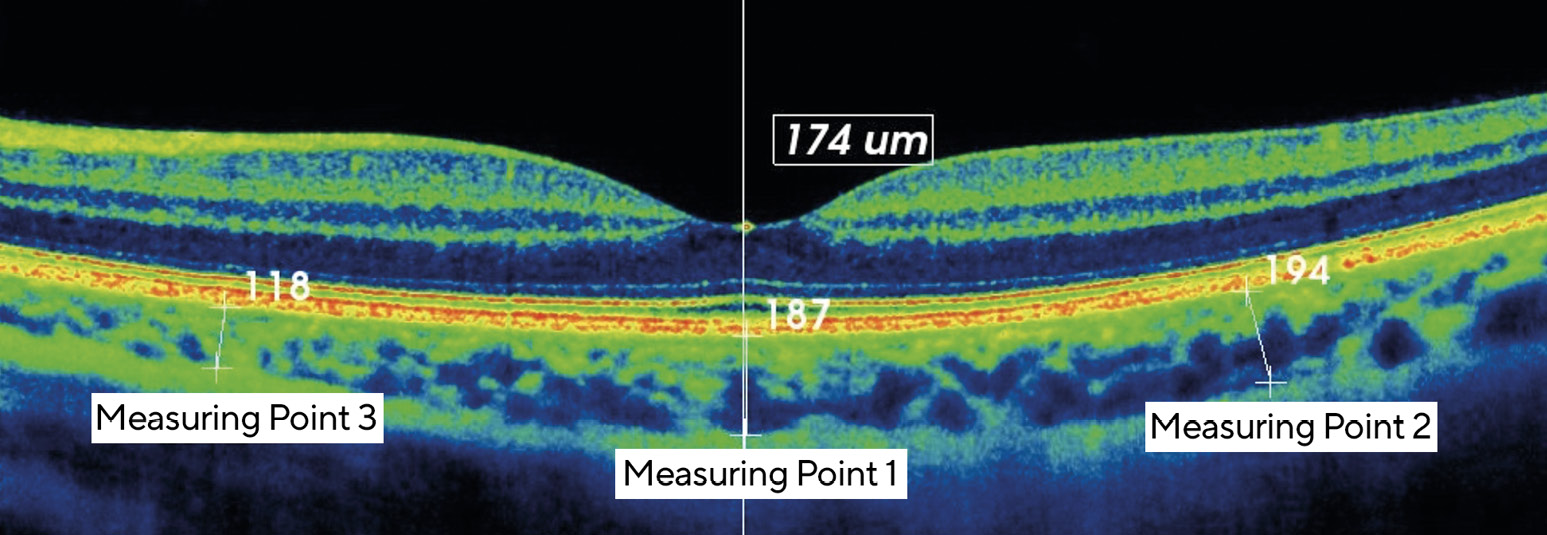
The choroidal thickness was determined by means of OCT, using a 3D-OCT 2000 (Topcon Corporation, Tokyo, Japan). For this purpose, a high-definition scan image (line scan) was made along a horizontal axis through the foveola and a subfoveal, a nasal and a temporal measuring point (each with a 2 mm separation from each other) were determined on each scan image. The actual measurement of the choroidal thickness was carried out by determining the distance between the retinal pigment epithelium (RPE) and the transition from the suprachoroid to the innermost scleral layer at each measuring point (Figure 1). The measurement was carried out on the OCT tomogram using an integrated manual measuring device. In order to trigger and maintain the process of accommodation over a sufficiently long period of time, a mobile phone with a high-definition display was attached to the side of the OCT device at a distance of 20 cm (demand of accommodation = 5.00 D) and a video was projected via the measuring beam path of the OCT using a semi-transparent mirror. The display had a luminance of between 10 and 50 cd/m2 depending on the video sequence, and the room brightness was between 10 and 100 lux during the accommodation phase.
The cold mirror used (FM03R, Thorlabs GmbH) reflects visible light (display of the smartphone) and transmits low-frequency light into the measuring beam path of the OCT. It was adjusted using a special holder (Figure 2) so that the test subject could view the video on the smartphone and an OCT measurement could be carried out at the same time. The examination procedure included a ten-minute phase to allow for accommodation relaxation (fixation on a distant object), a twenty-minute accommodative phase (accommodation on the smartphone display) and a further ten-minute relaxation phase. The first OCT scan was performed just before the accommodation phase, and a further six scans followed at five-minute intervals. We ended the accommodation stimulus as soon as the fifth scan had been taken (after twenty minutes of accommodation). Only one (randomly selected) eye was examined, the other being occluded with
an eye patch during the entire duration of the examination. The ametropia of the examined eye was corrected, if necessary, by a spherical or toric soft contact lens, the power of which corresponded to the ametropia at a vertex distance (VD) of 0.0 mm.
Our statistical analysis aims at answering to what extent (1) and in what time course (2) the process of accommodation leads to a change in the thickness of the choroid, whether myopia has an influence on these changes (3) and which changes can be observed during regeneration (4/5). Before the analysis, the series of measurements were checked for normal distribution using the Shapiro-Wilk test. Depending on the result, we then analysed the data with the corresponding statistical test. The calculation was carried out using the IBM SPSS software, with a significance level of α = 0.05.

Results
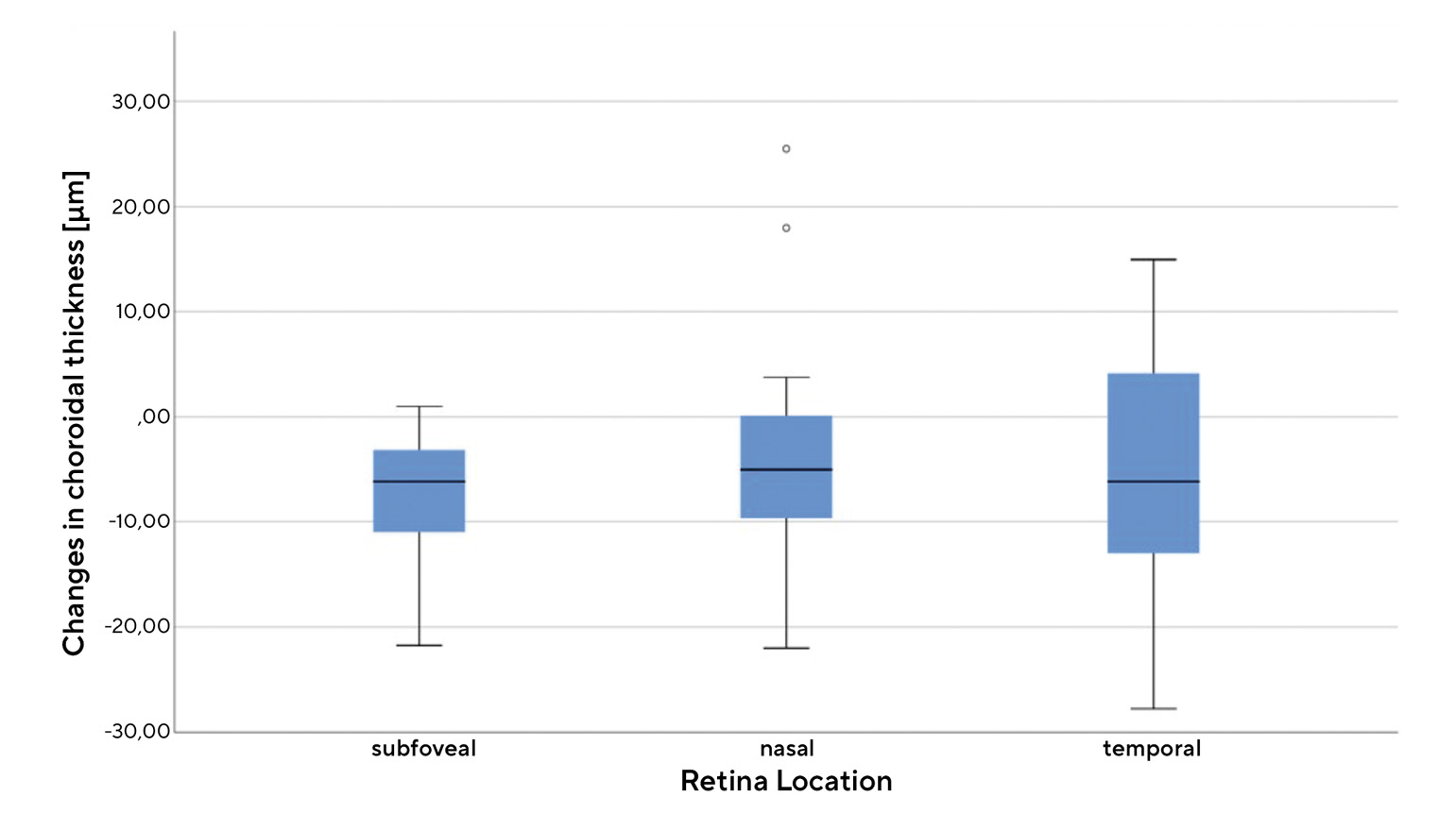
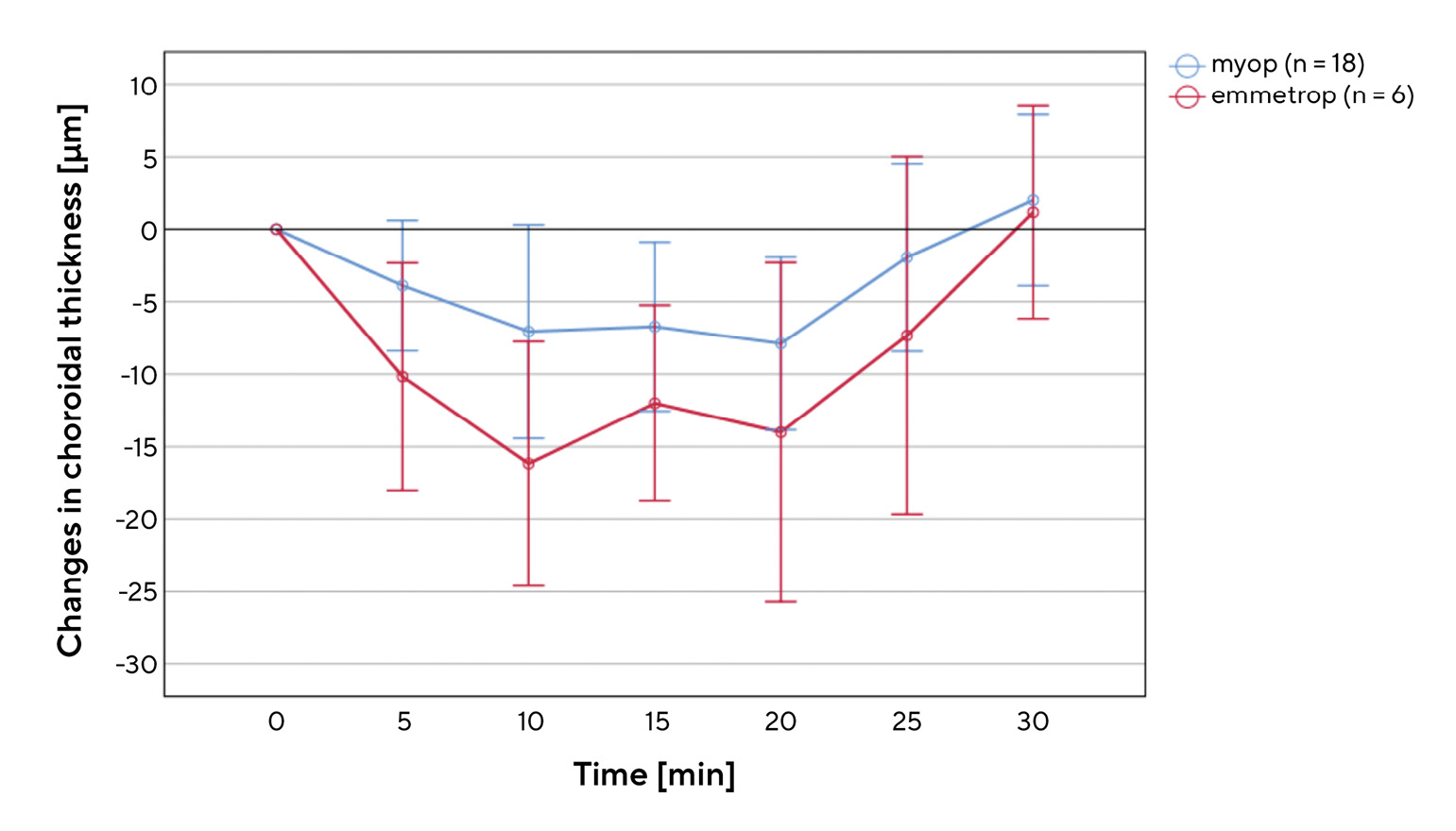
Figure 3 shows the average change in the choroidal thickness of all eyes included in the study within the 20-minute accommodation phase (difference values between the choroidal thickness at measurements 2-5 and the initial thickness). We measured a change in the subfoveal region of -8.06 ± 6.24 µm, in the nasal region of -4.0 ± 9.88 µm and in the temporal region of -5.29 ± 10.89 µm, which indicates a significant decrease in the choroidal thickness (Wilcoxon test, each p < 0.01). However, the standard deviations and the ranges shown in Figure 3 show that the measured values have a relatively high variability.Figure 4 shows the position and scatter for the temporal change in the subfoveal choroidal thickness in myopic and emmetropic eyes during and after the accommodation phase. During the first ten minutes of the accommodation phase, there was an approximately linear decrease in choroidal thickness in both groups. It then settled at a certain level and returned to the initial value within 10 minutes after the end of the accommodation phase.
The one-way ANOVA for repeated measurements (corrected according to Greenhouse-Geisser) assuming normally distributed data (Shapiro-Wilk test), showed that, at each measurement during accommodation, the subfoveal choroidal thickness of the two refractive groups differs significantly to the initial thickness (p < 0.01). However, a pairwise comparison (Bonferroni-corrected) showed no significant differences between the different measurements during the accommodation phase (p > 0.05 in each case). Five or ten minutes after the end of the accommodation phase, there is no longer any significant difference to the initial thickness (p = 1.00). The comparison between the change in the subfoveal choroidal thickness (during the accommodation phase) of both refractive groups with the Mann-Whitney U test for two independent samples showed that, on average, the choroidal thickness of emmetropic eyes decreased significantly more (p = 0.01) than that of myopic eyes (-13.08 ± 8.58 µm vs. -6.39 ± 6.06 µm). Nonetheless, the two refractive groups did not differ significantly (p = 0.80) regarding the regeneration of the choroidal thickness after accommodation (measurements 5-7). However, the error bars in Figure 4 show that there is a significant scatter in the measured values for both groups.
Discussion
The aim of this study is to obtain information on changes in the choroidal thickness during and after a prolonged accommodation task. Figure 3 confirms that the process of accommodation leads to a decrease in choroidal thickness, although this decrease varies depending on the region of the choroid. The statistical comparison and the results shown in Figure 4 also suggest that the decrease in thickness differs between myopic and emmetropic eyes and that it apparently does not take place spontaneously, but gradually. Both the thinning and recovery of the choroid needed approximately ten minutes to stabilise. The observations on the extent and the time course of changes in choroidal thickness during and after accommodation largely coincide with the few studies that have been published on this topic to date.8-10 The changes are small, but significant both within and between the two refractive groups. The gradual course of the changes visible in Figure 4 suggests that there is a certain dependency on the duration of the accommodation or on the time spent doing near work. This assumption can be easily reconciled with the fact that there is an obvious dependence on the distance of the close-up object observed or on the intensity of the associated accommodation stimulus. Studies with a demand of accommodation of +2.5 D8 or +3.0 D10 show no significant changes in thickness, while, on the other hand, the present study, with a demand of accommodation of +5.0 D, and the study by Woodman-Pieterse et al. with +6.0 D do show significant changes in choroidal thickness. However, the findings on the difference between the refractive groups of both studies are divergent. While the present study shows a smaller decrease in choroidal thickness in myopic eyes, Woodman et. al. found a stronger change in myopic eyes.9 One possible explanation for our results is that myopic eyes have a thinner choroid than emmetropic eyes due to stretching phenomena, meaning that they experience changes to a lesser extent upon accommodation. This assumption is supported, on the one hand, by the knowledge that the choroidal thickness decreases with severe myopia12 and, on the other hand, by the findings of the present study that the smallest decrease was found in the thinnest region of the choroid (nasal, Figure 3).
The decrease in the choroidal thickness observed in this study can possibly be explained by the fact that non-vascular smooth muscle cells (NVSMC) located in the choroid contract when there is an increase in the parasympathetic signal and fluid escapes from the “cavities” of the choroid.5 This assumption is supported by the findings of Woodman-Pieterse et. al., who found a correlation between the distribution of the NVSMC and the regional changes in choroidal thickness.10 Such an approach can at least be reconciled with the gradual decrease in choroidal thickness observed in our study if it is assumed that the fluid escapes from the tissue layers gradually rather than abruptly. It can also be argued that the adjustments in the choroid are caused by changes in the optical properties of the eye (aberrations, lag of accommodation, etc.) caused by accommodation. Studies show, however, that a retinal defocus of at least 3 D is necessary for a significant decrease in choroidal thickness,13 but such a defocus goes far beyond the deterioration of the retinal image due to aberrations that are normally expected during accommodation. Furthermore, the changes in the optical aberrations that are typically caused by accommodation do not match the changes in the different regions of the choroid.10 The measuring method developed in this study, in which an object was projected into the measuring beam path of the OCT device via a semi-transparent mirror, proved to be suitable. With this setup, in addition to determining the thickness of the choroid, a number of other biometric measurements are conceivable during active accommodation, such as determining the radius or thickness of the lens of the eye or changes in the overall axial length. The evaluation of the measurement procedure showed that neither wearing a soft contact lens nor the cold mirror had a significantly negative influence on the quality of the OCT scans or on the data collected on the choroidal thickness.
The strength of the present study lies in the development and testing of a cost-effective measurement method that allows for continuous accommodation and, at the same time, a sequence of OCT scans over a longer period of time. The relatively short repetition rate both during and after the active phase of accommodation should also be emphasised. One of the possible points of criticism of this study is that the OC tomograph used here sometimes reached its limits when displaying the outer border of the choroid and that the determination of the choroid thickness had to be done manually. This and the individual differences between the test subjects explain the relatively wide variation in the measured values for the accommodation-related changes in thickness (Figures 3 and 4) and that the reliability of the above statements depends on the multiple repetitions of the measurements. Further points of criticism would be that myopic and emmetropic eyes were included in unequal numbers, that the age range of the test subjects was limited and that the study does not contain any group of test subjects with a higher degree of myopia. This is mainly due to the fact that the aim of this study was primarily the development of a suitable measurement setup as well as obtaining first findings, in the spirit of a pilot study. In particular, the statements on the differences between myopic and emmetropic eyes should therefore be interpreted with appropriate caution. If the observed differences are confirmed in studies with a larger sample size and improved measurement technology (e.g., swept source OCT with enhanced depth imaging and analysis software), conclusions could then be drawn on the influence of the choroid on the axial length growth of myopic eyes, and the mechanisms of myopia genesis could be better understood.
Conclusions
The results of the present pilot study show that the process of accommodation leads to a decrease in the thickness of the choroid and that the choroid returns to its original thickness after the accommodation phase during which the eye was doing near work. The purpose-built setup developed to project a smartphone display into the measuring beam path of the OCT device proved to be suitable for stimulating accommodation over a sufficiently long period of time while, at the same time, performing repeated OCT scans. The results also suggest that the process of accommodation in emmetropic and myopic eyes is associated with different biometric changes in the choroid.
Conflict of interest
The authors declare that there is no conflict of interests regarding the methods and devices mentioned in the article.
COE Multiple Choice Questionnaire
The publication "The influence of accommodation on choroidal thickness: a pilot study" has been approved as a COE continuing education article by the German Quality Association for Optometric Services (GOL). The deadline to answer the questions is December 1st 2022 and only one answer per question is correct. Successful completion requires answering four of the six questions.
You can take the continuing education exam while logged in. Users who are not yet logged in can register for ocl-online free of charge here.
