MGD Treatment using Intense Pulsed Light and Low Level Light Therapy
Purpose: Primary objective of this study was the assessment of dry eye related subjective and objective findings of MGD patients prior and after IPL and LLLT treatment.
Material and Methods: 38 subjects with diagnosed MGD were enrolled in a prospective single center study in an optometric practice based in Wil (Switzerland). All subjects underwent IPL / LLLT treatments every 14 days from baseline until four treatments were conducted. OSDI, BCVA, IOP, NIKBUT, osmolarity, lower lid margin appearance and meibography data were captured at baseline before the first IPL/ LLLT treatment and 14 days after the last treatment.Pre and post OSDI score, NIKBUT and osmolarity of the right was statistically analyzed using Wilcoxon rang sum test for matched pairs.
Results: OSDI score decreased significantly from 32 to 14 (p = <0.001). NIKBUT values did not increased significantly from 11.4 ± 12.4 s to 11.7 ± 5.9 (p = 0.498). Osmolarity increased significantly from 322.2 ± 12.6 mOsm/L to 327.9 ± 9.6 (p = 0.006). 26 of 38 subjects (68.4 %) were satisfied or very satisfied by this new form of therapy whereas 12 subjects (31.6 %) did not report a subjective change.
Conclusion: The combined IPL / LLLT therapy is a promising therapy form usable in optometric and ophthalmological practices. The majority of the subjects reported subjective improvements, which were not confirmed by objective findings. Due to the high reduction of sicca symptoms and the high degree of satisfaction with the IPL / LLLT treatment, it can be stated, that an IPL / LLLT application is effective in MGD patients.
Introduction
Meibomian gland dysfunction (MGD) is considered as the main reason of dry eyes1 with a prevalence of 21.9 % in subjects aged 40 years and older.2 The disease can be described as a functional disorder of the meibomian glands. The main cause of MGD is an obstruction of the excretory ducts as well as a quantitative and/ or qualitative change in glandular secretion. It occurs diffusely because a majority of the glands are affected. The subjective sensation of an eye irritation can also be present for a positive diagnosis of an MGD.1 Most therapies require a daily treatment. This can be challenging for MGD patients, hence an excellent compliance is necessary to ensure a better quality and longer lasting effect of the therapy. A novel form of therapy for MGD patients is the Intense Pulsed Light application (IPL). An optometrist or ophthalmologist executes the complete IPL treatment, as it cannot be performed by the patient at home. If necessary, the efficiency of the IPL application can be combined with Low Level Light Therapy (LLLT).
IPL find its origin in the dermatological treatment area for facial telangiectasia, facial rosacea, and pigmented lesions or for hair removal.3 Estimated 5% of the global population is affected by rosacea4 while 80% of them have an accompanying MGD.2 In 2002, Toyos incidentally observed an improvement in signs and symptoms in MGD and dry eye disease (DED) patients receiving IPL treatment for facial rosacea. 5,6 IPL devices emit a broad-bandwidth, non-coherent and polychromatic light using a wavelength spectrum of 500-1200 nm with different pulse durations for selective thermal damage of the treated target.7,8 A chromophore located within the skin absorbs the radiant heat generated in the body cell.9 LLLT is used for skin treatment with red and near infrared wavelength ranges. While many forms of laser therapy apply high amounts of radiation energy, LLLT uses a "Low-Level" of radiation energy.10 The most important medical applications of LLLT are the reduction of pain and inflammation, the promotion of tissue repair and regeneration of various tissues and nerves, and the prevention of tissue damage. The metabolism in the stem cell is increased.10,11,12
Until 2020 only one study was available that described the efficacy of IPL application in combination with LLLT technology in MGD patients in the US.13 The mentioned study was a retrospective chart review including 230 patients at three sites, who underwent a combined IPL/ LLLT treatment using the EPI-C Plus/Eye-light® device. The parameters Ocular Surface Disease Index score (OSDI) and MGD grading resulted in a significantly decrease and tear break-up time nearly doubled after the treatment. A 1-grade or greater reduction in MGD classification was observed in 70 % of eyes, with 28 % of eyes having a 2-grade or greater reduction.13 Currently published studies14,15 confirmed Stonecipher´s study results. Pérez-Silguero found out that the positive treatment effect decreased during a twelve months post treatment follow up time, but OSDI was still better in comparison to baseline. A prospective study has been conducted in Switzerland to investigate if the retrospectively gained results of Stonecipher can be confirmed. Primary objective of this study was to investigate the subjective evaluation before and after a complete IPL/ LLLT application consisting of 4 sessions and the assessment of the OSDI score, Non-Invasive-Keratograph Break Up Time (NIKBUT) and osmolarity.
Method
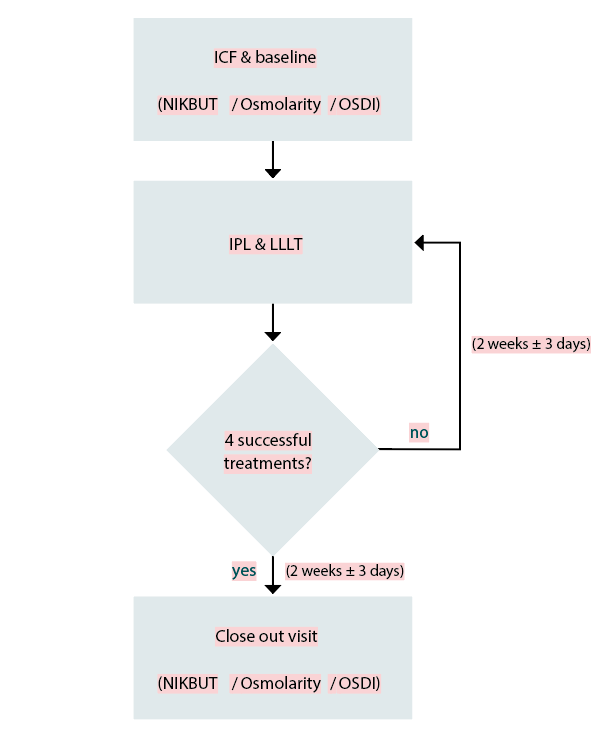
This prospective, longitudinal and univariate study was approved by the Ethics Committee of Eastern Switzerland (EKOS). The principles of the Declaration of Helsinki were strictly followed throughout the study. The complete study was conducted at the site of Bischof Optik AG in Wil (SG), Switzerland. A total of 38 subjects gave informed consent to participate in this study. Inclusion criteria were the age of 18 years, the diagnosed MGD and an OSDI score above 13. Potential subjects were excluded if theysuffered from Sjögren's syndrome or if they showed a history of any surgery of the anterior eye segment. Further exclusion criteria included: regular wear of contact lenses (CL are worn > 2 days / week all day), dermatological treatment of the face, permanent make-up on eyelid edges, epilepsy, pregnancy, taking photosensitive drugs, suspicion of skin lesions in the areas to be treated, metallic, electrical or acoustic prostheses, no ocular sensation, implanted pacemaker, cardiac arrhythmia, untreated high blood pressure and diabetes.
Clincal examinations
Demographics, medical and ocular history, best corrected visual acuity, intra ocular pressure (IOP), NIKBUT16, osmolarity, lower lid margin appearance, meibography according to the visualized grading scale of Arita available in the Keratograph 5M (Oculus Optikgeräte GmbH, Wetzlar, Germany) and OSDI score17 were captured. Table 1 shows the used instruments for the performed assessments. One measurement was conducted per eye. The examination findings were collected at the baseline visit prior to the first IPL/ LLLT treatment and at the exit visit 14 days +/-3 days after the last treatment. Ocular surface, eyelids and eyelid margins were assessed by using a slit-lamp regarding eyelid margin morphology and obstruction of the meibomian glands. It was documented whether the above-mentioned signs were predominant or not. Visual acuity and IOP were measured for control purposes only. Objective determination of NIKBUT, OSDI and meibography were conducted using the multifunctional topographer. Using the osmometer (TearLab, TLC Vision Corporation, USA), osmolarity was measured as an etiological factor for dry eye18 and as a fundamental component of the homeostasis of the tear film.
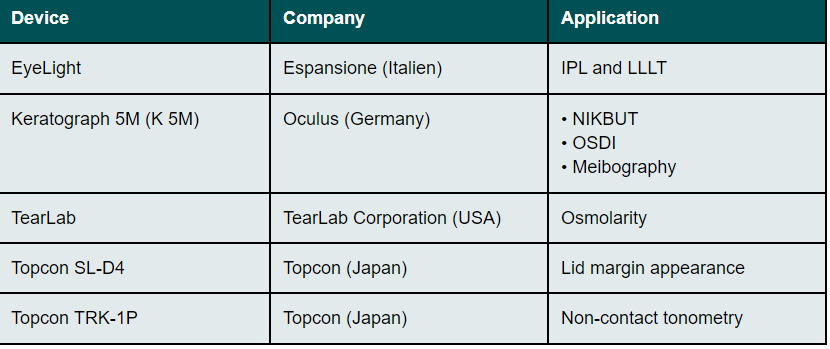
IPL treatment
All subjects were treated with the IPL device EyeLight (Espansione, Italy) and underwent combined IPL and LLLT therapies every 14 days +/-3 days from baseline until four treatments were conducted. The combination of IPL and LLLT in one treatment session is advantageous. The used device is the first one which integrates both applications. First, the IPL therapy was performed on both eyes with the handheld device, followed by the LLLT application using a face mask with inbuilt LEDs (Figure 1). Following the given instructions, the subjects were placed on a chair and leaned back until the head rests on the back of it. The eyes were covered with protective safety goggles according to the manufacturer’s recommendation. Prior to the first measurement, the investigator had to select the “Meiboscale (Meibomian gland atrophy) on the IPL device (Figure 2). An automatical determination of the skin pigmentation using the included Fitzpatrick scale was conducted at the treatment site using the handheld IPL device with an inbuilt 600 nm filter.19 Five IPL impulses, each with a duration of 10 ms were applied on each eye side. One impulse was placed at the temple area, one horizontally below the lower lid and three vertically below the lid according to the manufacturer’s recommendation. After the IPL treatment, the IPL flash on the handset was replaced with the LLLT mask (Figure 3). The LLLT mask was placed on the subject´s face and the subject was instructed to close the eyes. The subject closed their eyes and the mask started to emit light. After 20 minutes, the LLLT treatment was completed and the mask was removed from the subject's face. At the end of the exit visit the subjective satisfaction was determined by asking: "How satisfied are you with the result of the IPL / LLLT application?” As possible answers, the test persons could choose between "very satisfied", "satisfied", "no change" and "deterioration".

Statistical methods
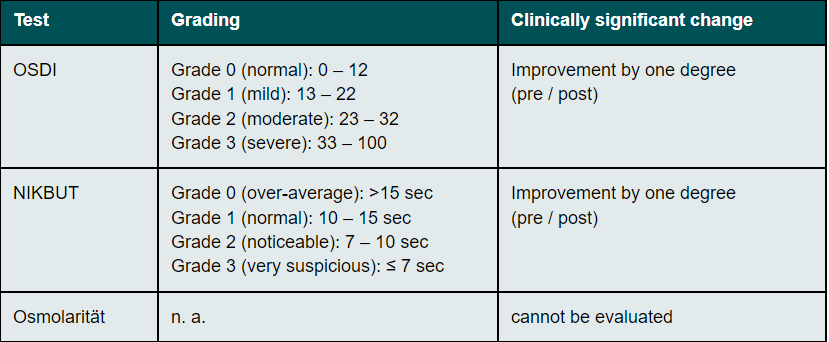
Statistical analyses were performed using SPSS Statistics 25.0 (IBM, Armonk, New York, USA). Determination of the sample size was not possible due to missing pilot data. Therefore, a sample size calculation based on assumptions (α 0.05, power 0.8, effect size -0.51, 10% drop out) was performed prior study start using G*Power.20 Pearson correlation showed a good accordance of results between the right eye (OD) and the left eye (OS). Hence only the OD results are shown. The Shapiro-Wilk test was used to test normal distribution of the data. Pre and post OSDI score, NIKBUT and osmolarity data were analysed using the non-parametric statistical hypothesis Wilcoxon signed-rank test due to missing normal distribution. The test was used to compare the clinical outcomes prior and post IPL/ LLLT treatment. P values less than 0.05 (α=0.05) were classified as statistically significant. The clinical importance orientated at the common gradings shown in table 2.


Results
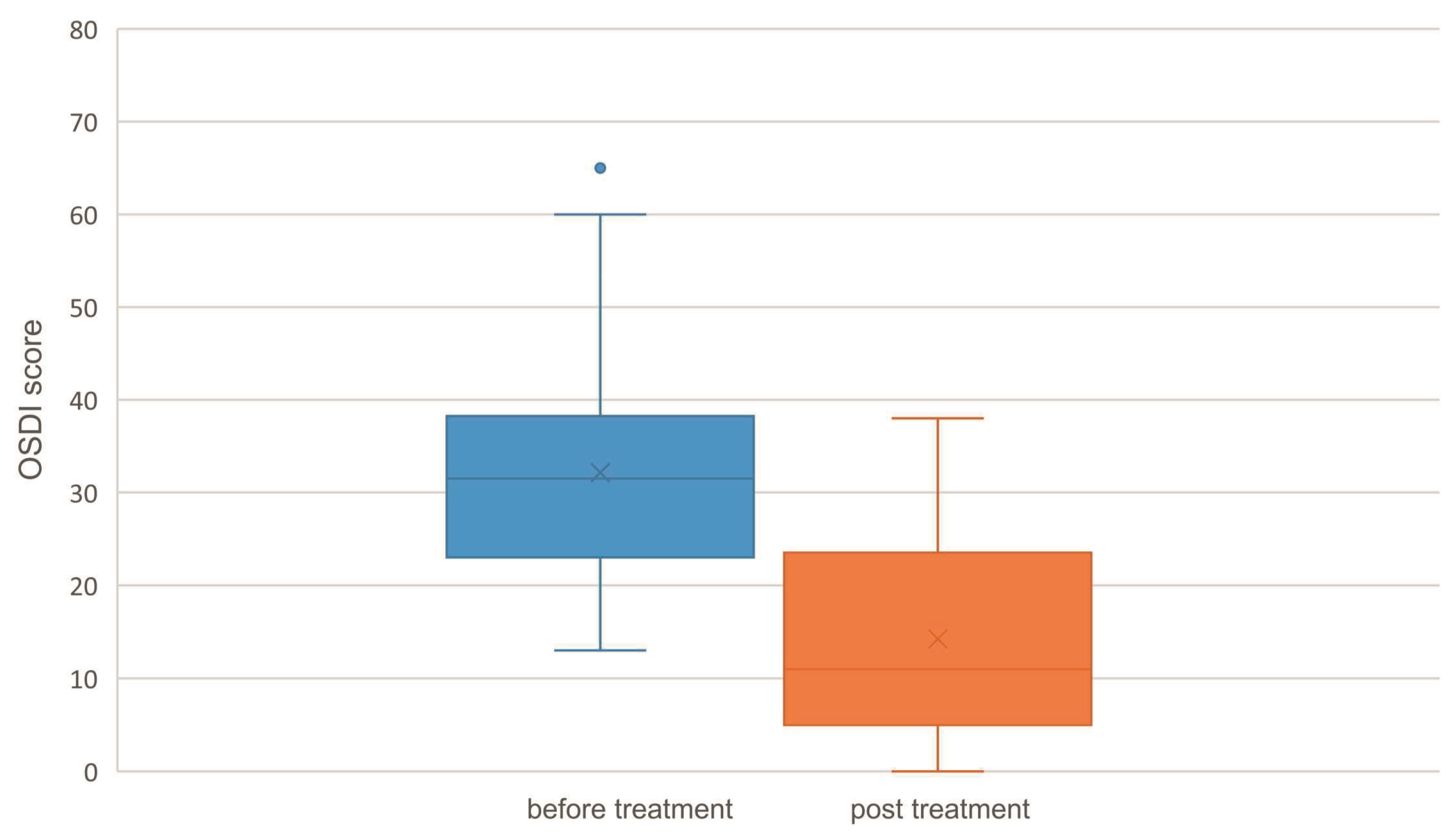
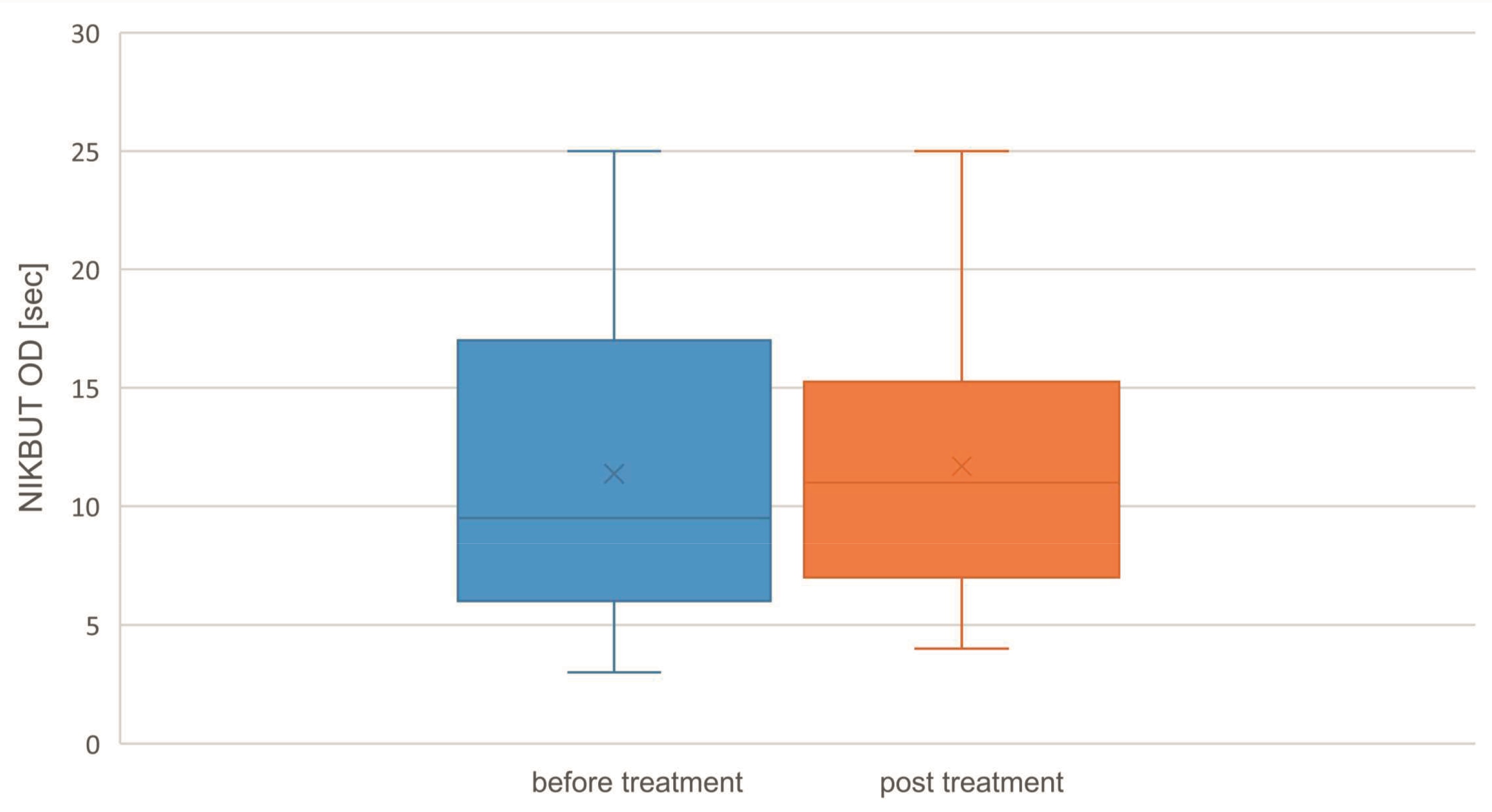
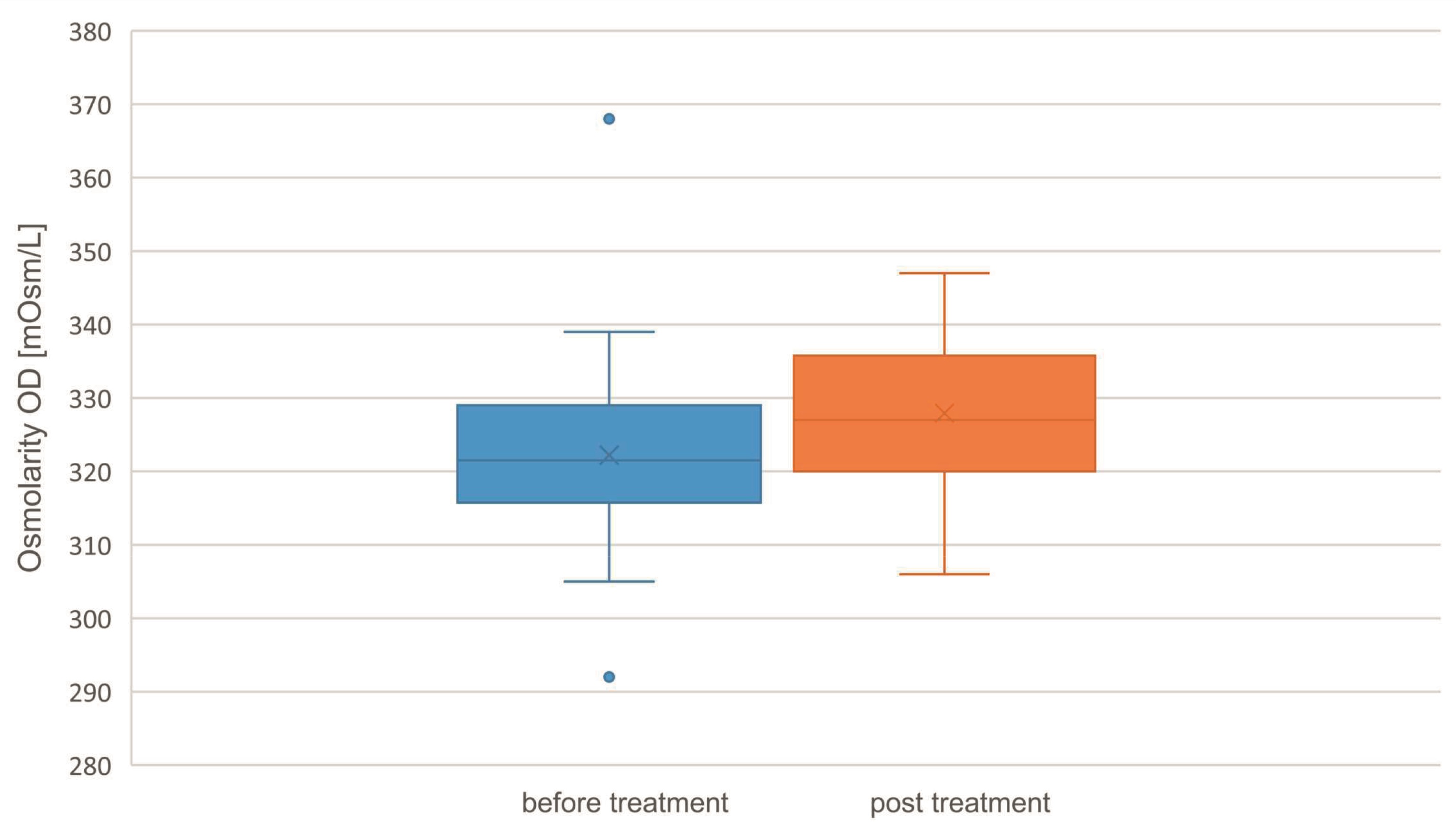
Thirty-eight subjects (27 women, 11 men) with a mean age of 54.2 ± 12.2 years were enrolled in this clinical trial. OSDI score, NIKBUT and osmolarity were compared using the initial value (pre) and the final value (post). All results are graphically displayed in figures 4 - 6. The OSDI score decreased significantly from 32.2 ± 12.4 to 14.2 ± 10.4 (P = <0.001). NIKBUT values increased not significantly from 11.4 ± 12.4 s to 11.7 ± 5.9 s (P = 0.498). The osmolarity increased significantly OD from 322.2 ± 12.6 mOsm/L pre to 327.9 ± 9.6 mOsm/L post (P =0.006). 26 of 38 subjects (68.4 %) rated the treatment itself as beneficial. 21 of the 38 subjects (55.3 %) were satisfied using this novel therapy and with the result of the IPL / LLLT treatment and five subjects (13.1 %) were very satisfied with the result. 12 subjects (31.6 %) did not report a subjective change.
Discussion
The OSDI score was considered to be the primary endpoint in this study. The subjective symptoms in everyday life can only be assessed by the subject. Therefore it can be assumed that an improvement in the subjective sensation of the test person can be rated higher than the objective changes. The mean OSDI score has been reduced from 32.2 (median 31.5) points prior treatment to 14.2 (median 11) points post treatment. This result shows a clinically relevant and statistically significant (P = <0.001) improvement in dry eye symptoms. To assess the clinical significance, the OSDI classification was compared before and after the IPL/ LLLT application. The classification is reduced from moderate to mild suggesting that the clinical significance is high (Table 5). The hypothesis "A complete IPL/ LLLT application (4 sessions) reduces the OSDI score" can thus be statistically and clinically confirmed.
The study showed that a combined 4 cycle IPL / LLLT treatment results in a mean increase in NIKBUT by 0.3 sec (median 1.5 sec) (P = 0.498). The statistic shows that there is no significant difference between the NIKBUT pre and post treatment. On this basis, an improvement of this parameter could not be confirmed. Other studies with similar designs show an increase in break-up times ranging from 1.2 s to 8.83 s.21,22,23,24,25,26,27,28 The range is comparable with the data in the conducted study. Although the increase of tear film break up time was not as high as demonstrated in previous studies14,23, it indicates a clinical benefit for the treated subjects.In addition to the observed improvements, the osmolarity of the tear film has increased due to the IPL / LLLT application and thus deteriorated in relation to dry eye. Mean osmolarity has changed from 322.2 mOsm/L to 327.9 mOsm/L (median 321.5 mOsm/L to 327.0 mOsm/L). The hypothesis "These findings indicate that a complete IPL/LLLT treatment does not improve tear osmolarity, but it does improve subjective responses as assessed by OSDI”.
A clinical relevant improvement or deterioration cannot be confirmed. However, compared to results from other studies, the obtained data are similar with one exception.25,27,28,29 In these studies, the osmolarity changes are described between -6.0 mOsm/L (reduction/ improvement) and 24.2 mOsm/L (increase/ deterioration). Only the recently published study by Perez-Silguero14 showed a decrease in osmolarity by 52.8 units from 331.8 mOsm/L to 279.0 mOsm/L. Although the subjective uncomfortable ocular feeling of dryness was improved no objective tests showed clear changes. Only the median NIKBUT values showed slight improvements.In addition, the eyelid margins and meibography status were assessed. Owing the fact that the meibomian gland atrophy is irreversible, all subjects showed no meibography improvements with a mean grade of 1.05 before and after IPL / LLLT application. In conclusion, the IPL/ LLLT therapy could be classified as beneficial for an optometric practice by the subjective response of the treated test persons. This was claimed by 26 of 38 test persons and corresponds to 68.4 % of the subjects in the tested cohort. In summary, the statistical analysis of NIKBUT and osmolarity both revealed no statistically significant improvements. The OSDI improved statistically highly significant.
Conclusion
An IPL / LLLT application in MGD patients can certainly be considered a sensible form of therapy. For a higher success of treatment, the selection of subjects needs to be carried out more specifically. This means that only subjects with MGD grade 2-3 (minimal to moderate) are evaluated according to the known grading of clinical MGD staging.30 Probably the treatment results would be even better, because the DEWS II Report shows that advanced sicca, which includes MGD grade 3-4, are mostly mixed forms between hypovolemic and evaporative. Thus, IPL / LLLT therapy alone cannot provide total success. However, the shown classification changes provide a good basis to discuss the effectiveness of IPL / LLLT application in MGD patients. Due to the high reduction of sicca symptoms and the high degree of satisfaction with the IPL / LLLT treatment, it can be stated that an IPL / LLLT application is effective in MGD patients. However, a cycle of 4 IPL / LLLT treatments alone, conducted under the described methodical aspects, seems to be not effective to cause objective improvements.
Conflict of interest
The authors declare that there is no conflict of interests regarding the methods and devices mentioned in the article.
