Myopia treatment with low-dose atropine in clinical practice
Purpose: The aim of the study was to determine the therapeutic effect of low-dose atropine for inhibiting myopia progression in clinical practice outside of controlled clinical trials.
Material and Methods: We performed a retrospective analysis on data collected on a regular basis (refraction, axial length) pertaining to 79 children treated with atropine and 100 untreated children over an observation period of 12 months.
Results: Our findings show that myopia progressed in both groups with highly variable progression rates. A two-way ANOVA for age and atropine effects on axial length growth revealed that axial length growth rates decline with age (p < 0.0001) and that atropine has a significant inhibitory effect on axial length growth (p < 0.0015) independent of age. However, this effect showed on average only a 0.08 mm inhibition rate per year (28 %) as compared to the growth rate in the untreated children, meaning that its magnitude is quite small. Furthermore, the effect of atropine on the refractive power was not significant.
Conclusion: We found that, in this clinical setting, the effects of atropine were not distinctive and of only minor clinical relevance. The results of this study show that the beneficial effects of a treatment with 0.01% atropine eye drops may not be apparent in every individual case, information that should be communicated to the parents of myopic children and general ophthalmologists.
Introduction
Myopia is caused by a disparity between the refractive power and the length of the eye.1 As a result, the distant focal point lies in front of the retina in the vitreous humor and a blurred image is created on the retina.1 Axial myopia (longer eye globe) is the most common type of myopia in children and is due to excessive axial length growth.2 As the axial length increases, so does myopia and the risk of more serious diseases in old age, such as glaucoma, retinal detachment and myopic macular degeneration, especially with high degrees of myopia.3,4,5,6 Myopia usually begins during childhood7, typically at the beginning of primary school. It progresses during adolescence and this progression may continue into young adulthood. An increasing prevalence of myopia can be observed not only in the newly industrialised East Asian countries, but also in Europe.8
In recent years, strategies and treatment options that inhibit the progression of myopia have been developed, including soft and rigid multifocal contact lenses9,10, orthokeratology lenses9,11, peripheral defocus spectacle lenses12,13 and atropine.14,15 The effectiveness of low-dose atropine treatments with comparatively few side effects has been proven in randomised clinical studies under strictly controlled conditions. 15-18 The effectiveness of atropine therapy in inhibiting myopia progression is very dose dependent.15 However, the side effects also increase with an increase in dose.19
It is known from other ophthalmic drop treatments, that the patient compliance, i.e. the extent to which the patient sticks to the treatment as agreed with the doctor, is not optimal.20,21 As a result, these treatments cannot achieve the same efficacy as shown in clinical trials.
The aim of this study is to evaluate the effectiveness of low-dose atropine in everyday clinical practice outside of controlled clinical trials. For this purpose, we retrospectively analysed the biometric data and the development of refractive power of myopic children, who are patients in our practice.
Patients and Methods
Over the past four years, we prescribed an atropine treatment on myopic children which consisted of applying one drop of a highly diluted atropine solution (0.01 %) in each eye, each night before going to bed for 5 days. This treatment scheme corresponds to the minimum requirement specified in the LAMP study.15 A stricter, and, therefore, more stressful drop schedule was not prescribed in order to maintain compliance of the young patients and their parents over a long period of time. Follow-up visits were scheduled to take place every 6 months. In each of the visits, refractive power and axial length were measured. The biometric determination of axial length was carried out using the IOL-Master 700 (Carl Zeiss Meditec, Jena, Germany). Subjective refraction was partly determined without the use of cycloplegic drugs. However, cycloplegic drugs were administered when children showed an obvious tendency to accommodation.
New glasses were prescribed if a change in refraction of more than 0.5 D was measured and a resulting increase in visual acuity was observed. The present study shows a 1-year analysis consisting of data from 115 children treated with atropine who had at least one follow-up visits. Refraction and axial length are routinely measured in our practice also in myopic children who do not receive atropine eye drops or any other form of myopia-inhibiting treatment. 212 untreated myopic children with at least two visits served as a control group for this study.
Since the actual time intervals between the visits were very different, only data from children who had a follow-up visit within a period of 9-15 months after the start of the treatment were statistically evaluated. This was usually the second or third visit after start of the treatment (1st visit). This range of 9-15 months is hereinafter referred to as the “12-month interval”. The growth rates (growth per year) were individually normalised from the actual time interval between these two visits to a 12-month period. Earlier or later check-ups of the same children were not included in the statistical analysis. Taking all of this into account, data of 79 atropine treated children, whose follow--up visit took place 12.7 ± 1.4 months after start of treatment, and 100 myopic children without treatment and a follow-up visit after 12.5 ± 2.1 months were analysed. For statistical analysis, a two-way ANOVA was carried out, considering the age of the children and the type of treatment (atropine treatment vs. no treatment).
The analysed data were collected during routinary clinical practice; no additional data were collected, or additional measures taken.
Results
The patient characteristics are shown in Table 1. Overall, 16.7 % of the children reported side effects; These were primarily burning eyes after administering the drops (9.7 %), dilated pupils on the day after having applied the drops the evening before (5.6 %), photophobia (5.6 %) and red eyes (5.6 %). One child (< 1 %) reported problems when reading text up close (15 cm) and another child had the feeling of having to blink more often and harder. We did not detect a clinically significant reduction in accommodation caused by the atropine treatment.
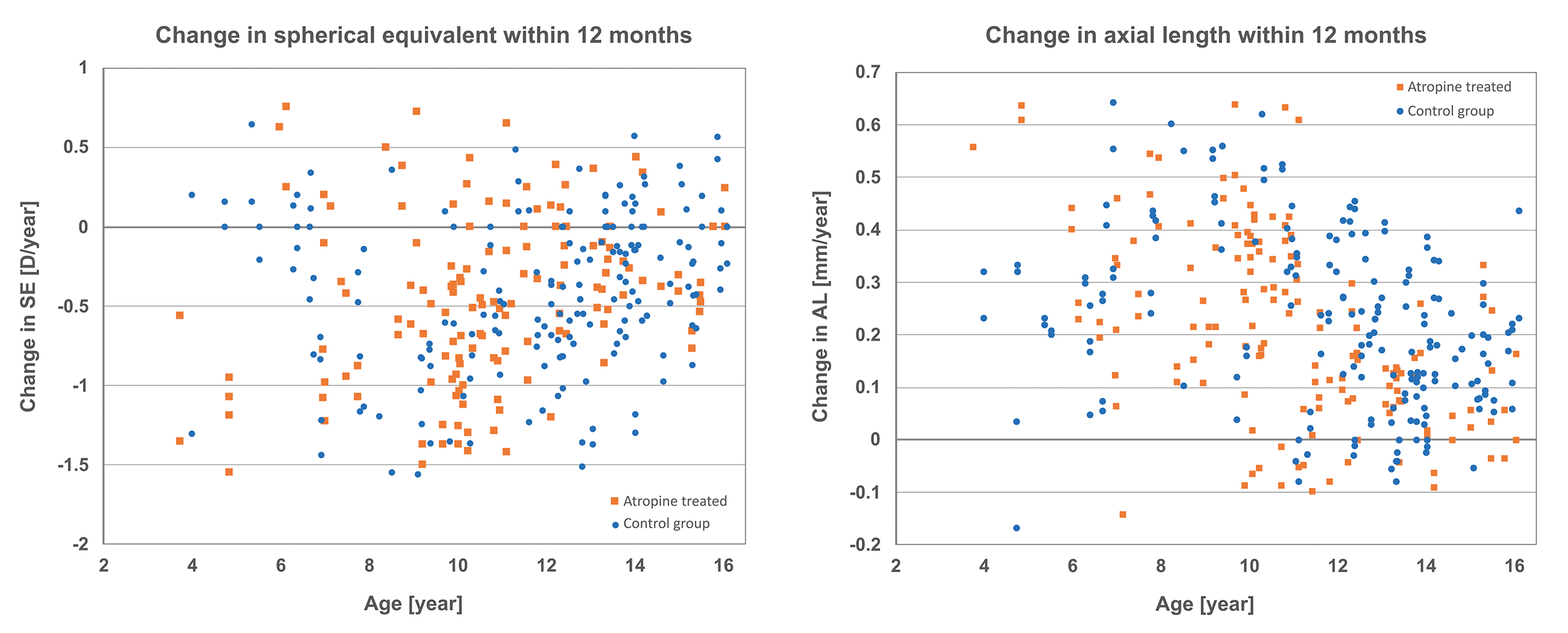
To evaluate the effects of low-dose atropin on myopia progression, Figure 1 shows the myopia progression rate (D/year) and the axial length growth rate (mm/year) for each eye.
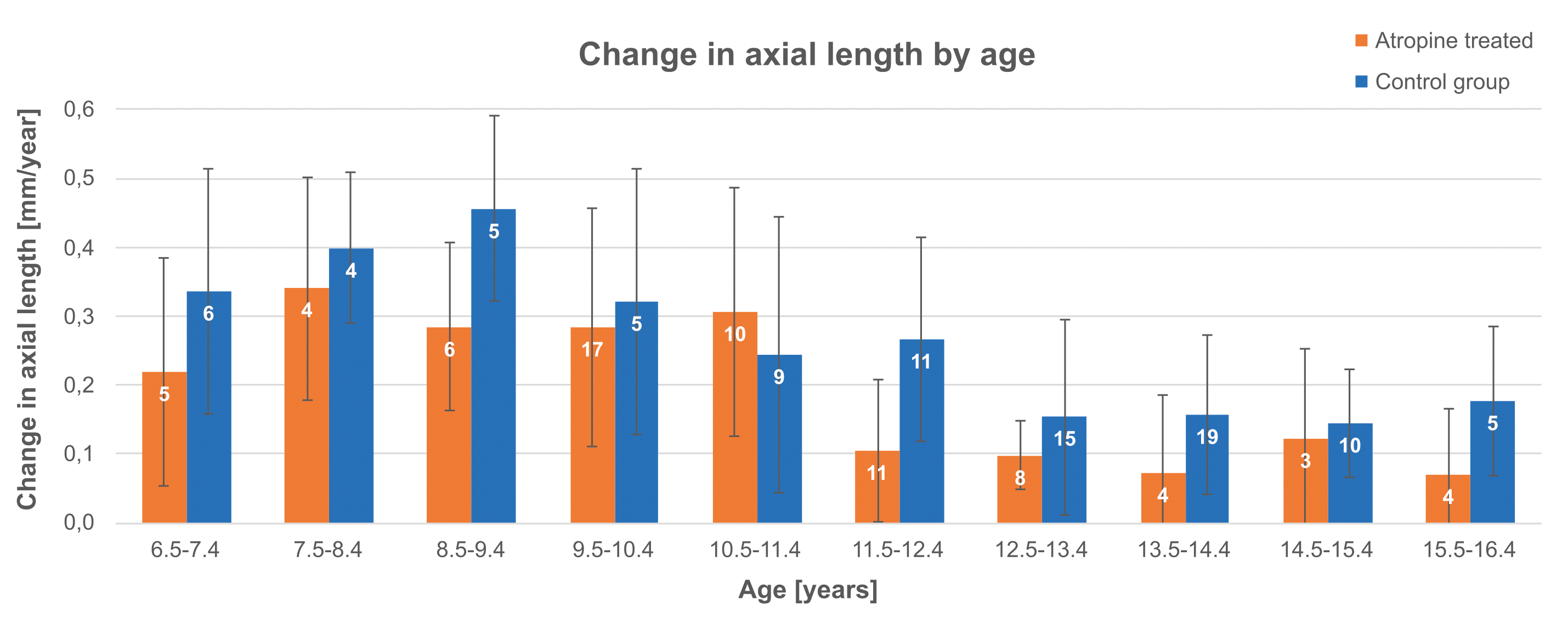
In Figure 2, the data of the children (6 years or older) of both treatment groups were divided into 10 age groups, to compare annual axial length growth only in children of the same age. The number of children under 6 years of age per group was too low and their data were, therefore, not included in Figure 2 nor statistically evaluated. A greater increase in axial length can be observed in the control group for almost all age groups. The two-way ANOVA shows that the annual growth rate decreases with age (p < 0.0001). Furthermore, our analysis shows that the atropine treatment inhibits axial length growth regardless of age (p < 0.0015). For each age group the difference in axial length growth of children who received atropine treatment compared to control group was calculated. On average, we found an inhibition of axial length growth of 0.08 mm (28 %) in the atropine treated group. The same analysis on measurement of refractive power (spherical equivalent) showed that the effect of atropine on refraction was not significant (p = 0.6).
If the children are grouped by degree of axial length growth, 51 % of the children treated with atropine and 47 % of the myopic children in the control group showed a mild progression of less than -0.5 D/year; 26 % of the children treated with atropine and 28 % of myopic children in the control group showed a moderate progression of -0.5 to -1.0 D/year; 23 % of the children treated with atropine and 25 % of the myopic children in the control group manifested a severe progression (more than -1 D/year).
Discussion
The data presented and analysed in this study do not derive from a randomised and controlled trial, but were collected during everyday clinical practice and therefore reflect the circumstances under which children receive atropine eye drops to treat myopia in Germany.
The effect of low-dose atropine (0.01 %) on myopia progression in clinical practice was, on average, not very distinctive. Statistically significant effects were only apparent when all patients together were considered, meaning that the intended inhibitory effect of the atropine treatment on myopia progression would not become noticeable in every individual case. This was also established by the Asian LAMP study by Yam et al.14,15
Our results showed an inhibition of axial length growth in children with atropine treatment that was independent of age. Li et al.22, on the other hand, found that eyes of younger children were less responsive to the atropine treatment.
Additionally, we solely found an atropine effect on axial length but not on refraction. This may be due to the fact that determination of refraction was not carried out consistently under cycloplegia. Brennan et al.23 proposed the “cumulative absolute reduction of axial elongation” to quantify the effect of therapies to treat myopia. The axial length is the more relevant value for estimating the individual risk of pathological changes due to a high degree of myopia, since these changes are directly related to the excessive axial growth of the eye globe.
The implementation of treatments with eye drops is subject to personal responsibility and, therefore, patient compliance can be low and very variable.20,21 In the retrospective analysis presented here, no control instrument was used to check compliance with the treatment. It was explained to the parents that consistent and regular use of the atropine drops was necessary in order to inhibit the myopia progression. However, random checks showed that in some cases up to 50 % less atropine than prescribed was ordered.
Common treatment options for myopia are presented in Figure 3. As in Figure 2, the average annual change in axial length (eye growth) after 1 or 2 years of respective treatment was plotted against the children’s age. The growth rates of the control groups of these studies are also shown (in blue); they represent the myopic (excessive) eye growth of the untreated children. The continuous grey line shows the axial length growth we extracted from the work of Truckenbrod et al.24 of children who are still emmetropic in adulthood (emmetropic eye growth). In the treatment of myopia in clinical practice, emmetropic eye growth could be used as a target for therapeutic efficiency.
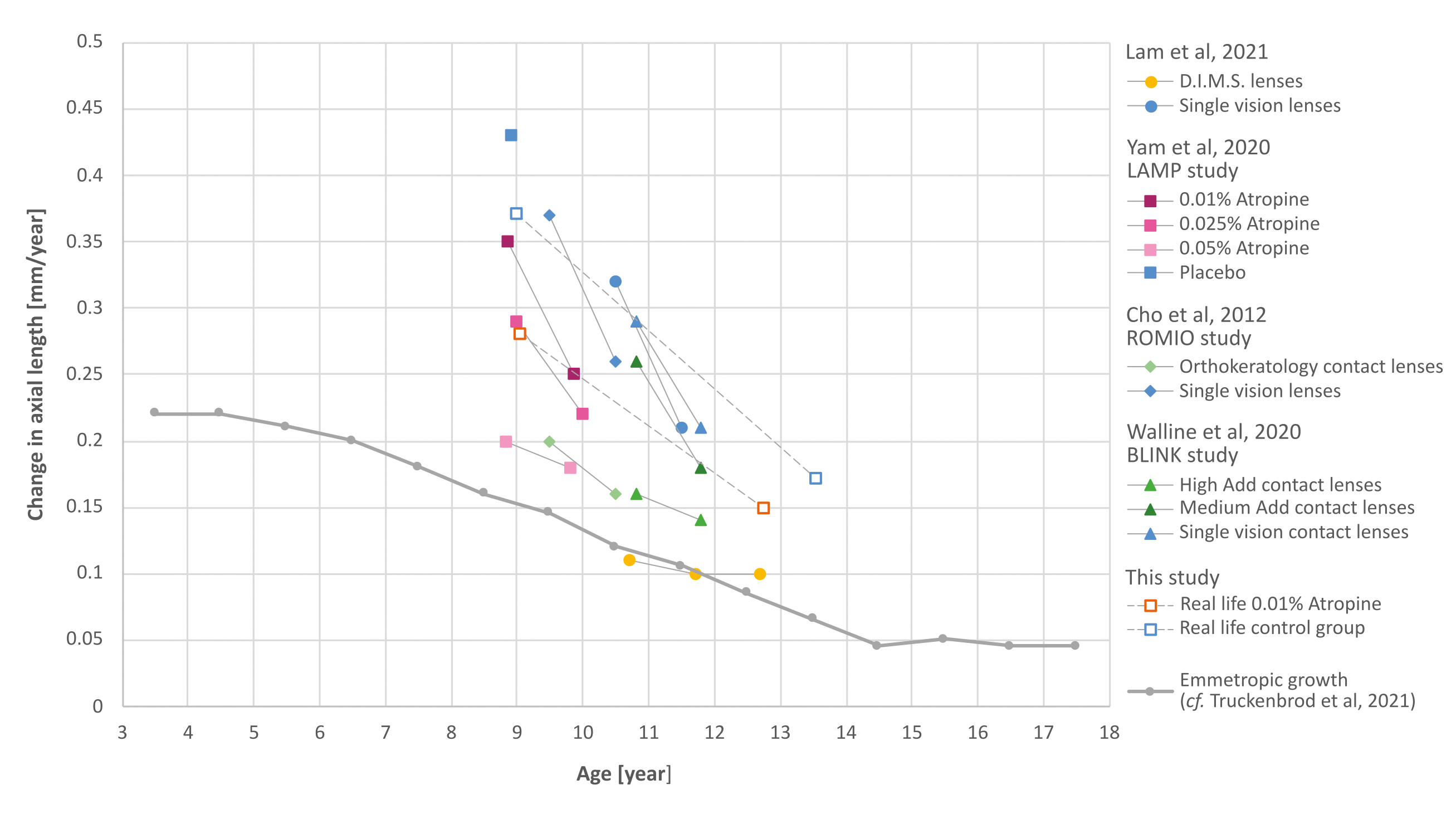
The data of the present study (empty squares) have been divided into two age groups for comparison. These were chosen so that they correspond to the age of the children in the LAMP study14,15 after the first year of atropine treatment. The atropine-induced growth inhibition found in the 12-month interval corresponds roughly to what was achieved in the Asian LAMP study only with a higher atropine dose (0.025 %).15 However, this statement must be put into perspective, in view of the significantly stronger myopic eye growth in the LAMP control group. Sanz-Diez et al.25 show that emmetropic eye growth is rarely found in the Asian population. Nevertheless, according to our analyses, emmetropic eye growth of Asian children hardly differs from that of German ones (adapted from Truckenbrod et al.24).
Similar results can be found for the treatment options of orthokeratology lenses (“OrthoK”) from the ROMIO study by Cho et al.11 and bifocal contact lenses from the BLINK study by Walline et al.10 It can be seen in Figure 3 that, in these clinical studies, the therapeutic efficiency of the orthokeratology lenses11 and that of bifocal contact lenses with high near addition10 are approximately the same as the therapeutic efficiency of the maximum dose of 0.05 % atropine in the Asian LAMP study.14,15 According to recent studies by Joachimsen et al.26, this dose exceeds the tolerance limit of long-term atropine treatment in German children.
The 1 to 3-year results of a first clinical study on new bifocal spectacle lenses with peripheral defocus (D.I.M.S.) by Lam et al.13 are remarkable. They show a comparatively high treatment efficiency after one, two and three years of treatment, respectively. The direct comparison with the results of the LAMP study14,15 is particularly interesting, since the patients come from the same ethnic group and the same socio-cultural background.
Conclusions
- Our retrospective analysis shows that a statistically significant but clinically weak effect of low-dose atropine on axial length growth can also be observed in a non-controlled and non-randomised study.
- However, under the given conditions, the effects were not recognisable in all patients, findings which should be conveyed to parents of myopic children in order to prevent excessive expectations.
- Alternative treatment options may inhibit the progression of myopia in the patient to a greater extent
Conflict of interest
The authors have no conflict of interest with regard to the methods and devices mentioned in the article.
