Progression of Reticular Dystrophy of the Retinal Pigment Epithelium
Purpose. This case report describes the progression of reticular dystrophy of the retinal pigment epithelium in a 61-year-old patient over a period of 11 years.
Material and Methods. A patient, now 61 years old, presented three times between 2013 and 2025 for a comprehensive eye examination. Based on indirect ophthalmoscopy and laser scanning ophthalmoscopy (Optos Daytona Plus, Nikon Co. Ltd., Japan), abnormalities in the retinal pigment epithelium (RPE) of both eyes were discovered during these examinations. The progression of these RPE changes is presented and discussed based on Optomap ultra-wide-angle imaging and mixed color view/fundus autofluorescence (FAF) as well as an OCT (Copernicus REVO OCT, Optopol Technology, Zawiercie, Poland) examination.
Results. During the initial consultation, discrete pigmentary changes were detected in both eyes, located peripapillary, macular, and in the central fundus.These changes were more pronounced in the right eye than in the left. Over the following 11 years, the pigmentary changes increased continuously, resulting in a picture of reticular dystrophy of the retinal pigment epithelium, which is one of the pattern dystrophies of the RPE.
Conclusion. A comprehensive eye examination often reveals asymptomatic findings that require regular monitoring or treatment, depending on their aetiology. Reticular dystrophy of the retinal pigment epithelium belongs to the group of pattern dystrophies of the RPE; since its visual prognosis is usually relatively good, treatment is not indicated in most cases.
Introduction
Reticular dystrophy of the retinal pigment epithelium (RPE) was first described by Swedish ophthalmologist Henrik Sjögren in 1950.1 Along with butterfly dystrophy, foveal vitelliform dystrophy, multifocal pattern dystrophy (simulating Stargardt‘s disease) and fundus puverulentus in adulthood, it is one of the pattern dystrophies of the RPE.2 These dystrophies belong to a heterogeneous group of macular diseases with pigment (lipofuscin) deposits in the RPE.2 They are often mutations in the human “RDS (retinal degeneration) gene” and the “peripherin gene.”3 The RDS/peripherin gene is crucial for the development of peripherin-2, a cell surface glycoprotein. This may be involved in the development and stabilization of the outer photoreceptor segments.4,5 Although pigmentary deposits occur in most cases in the macula, they can also manifest in peripheral areas of the retina. Pattern dystrophies are primarily inherited in an autosomal dominant manner.2 Although pattern dystrophies of the RPE are considered relatively rare findings in the literature 6, their incidence appears to be significantly higher.7,8 The reason for this is, firstly, that asymptomatic patients often do not undergo further examinations and, secondly, pattern dystrophies are often confused with age-related macular degeneration (AMD).7 There is little reliable epidemiological data available for pattern dystrophies. A regional study found that the incidence of vitelliform macular dystrophy in adults in Olmsted County, Minnesota, was between 1 in 7,400 to 1 in 8,200 diagnosed cases.9 In general, the visual prognosis for patients with pattern dystrophy of the RPE is relatively good, although pigmentary deposits in the macula can lead to reduced vision, choroidal neovascularization, and macular holes.2
Case description
Examination February 2013
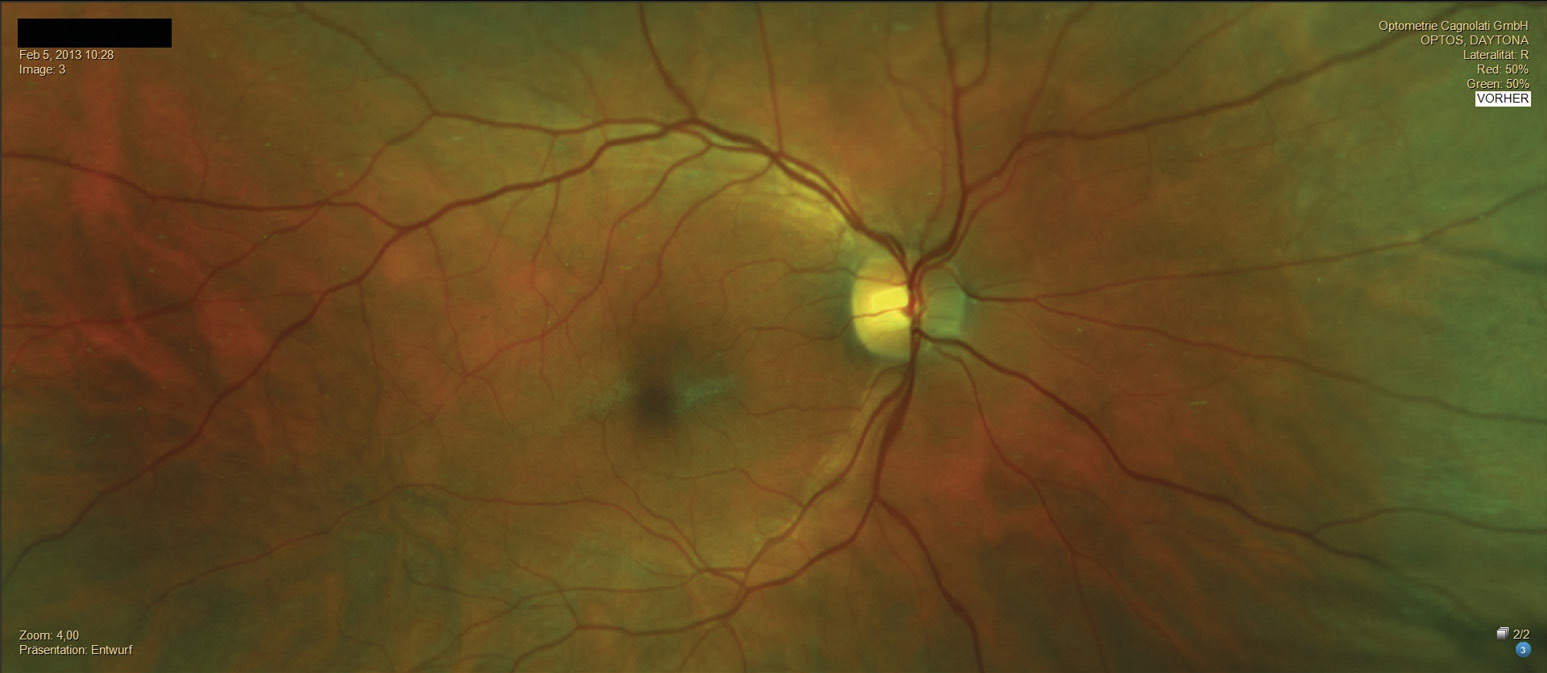
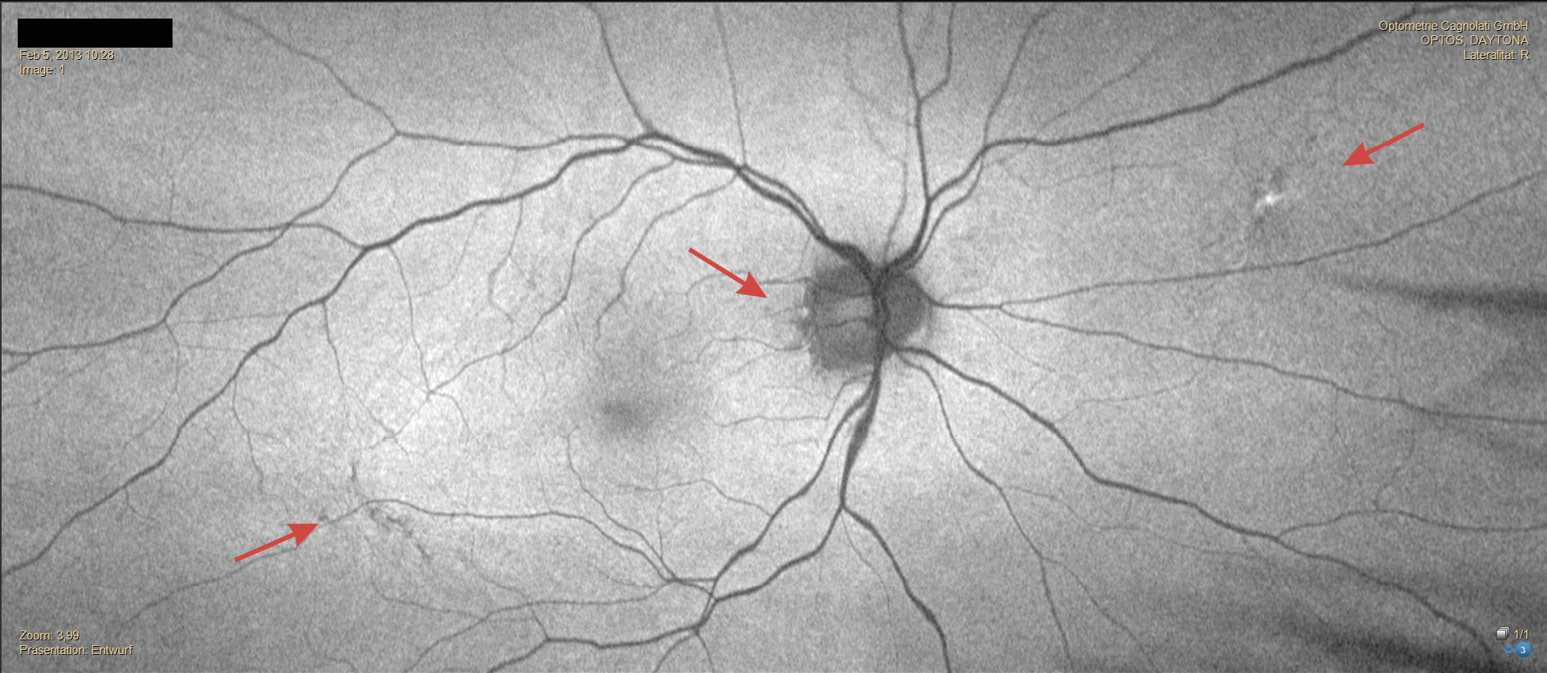
In February 2013, a 50-year-old patient presented for the first time for a comprehensive eye examination and new visual aids. The family history revealed age-related macular degeneration (AMD). The monocular and binocular refraction showed correction powers of OD: +1.50 D cyl −2.50 D axis 179° and OS: +1.00 D cyl −3.50 D axis 1°, with monocular visual acuities of 20/25 and binocular visual acuity of 20/20 p. Based on the measured monocular accommodation amplitude (OD: 2.5 D – OS: 2.3 D), a near addition of 1.25 D was determined. The examination of the anterior segment of the eye revealed no abnormalities. The crystalline lens showed no significant opacities in either eye. The posterior segment of the eyes showed a cup-to-disc ratio of 0.45 in the right eye and 0.4 in the left eye. The optic disc showed normal color and distinct margins in both eyes, and the macula was regular. Furthermore, discrete pigmentary changes were detected in both eyes, located peripapillary, macular, and in the central fundus.These changes were more pronounced in the right eye than in the left (Figure 1 and Figure 2). The IOP was 19 mmHg in both eyes [(11:22 a.m.), Topcon CT-800, Topcon Healtcare, Japan]. The C-20-5 visual field screening performed using frequency doubling perimetry [(Zeiss Humphrey FDT), Zeiss, Germany] was unremarkable. Due to the subjective asymptomatic nature and the very discrete pigment changes, we discussed the findings with the patient and agreed on regular follow-up examinations.
Examination in April 2016
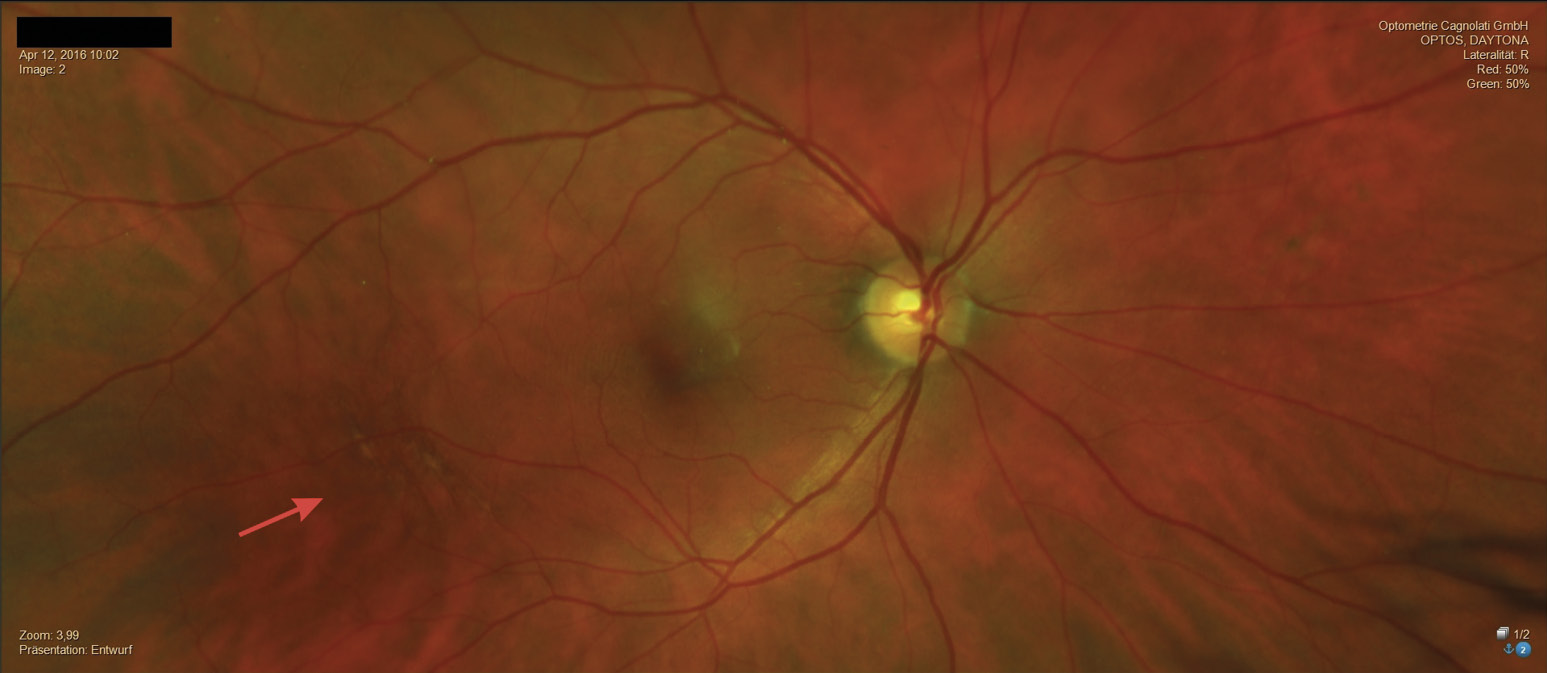
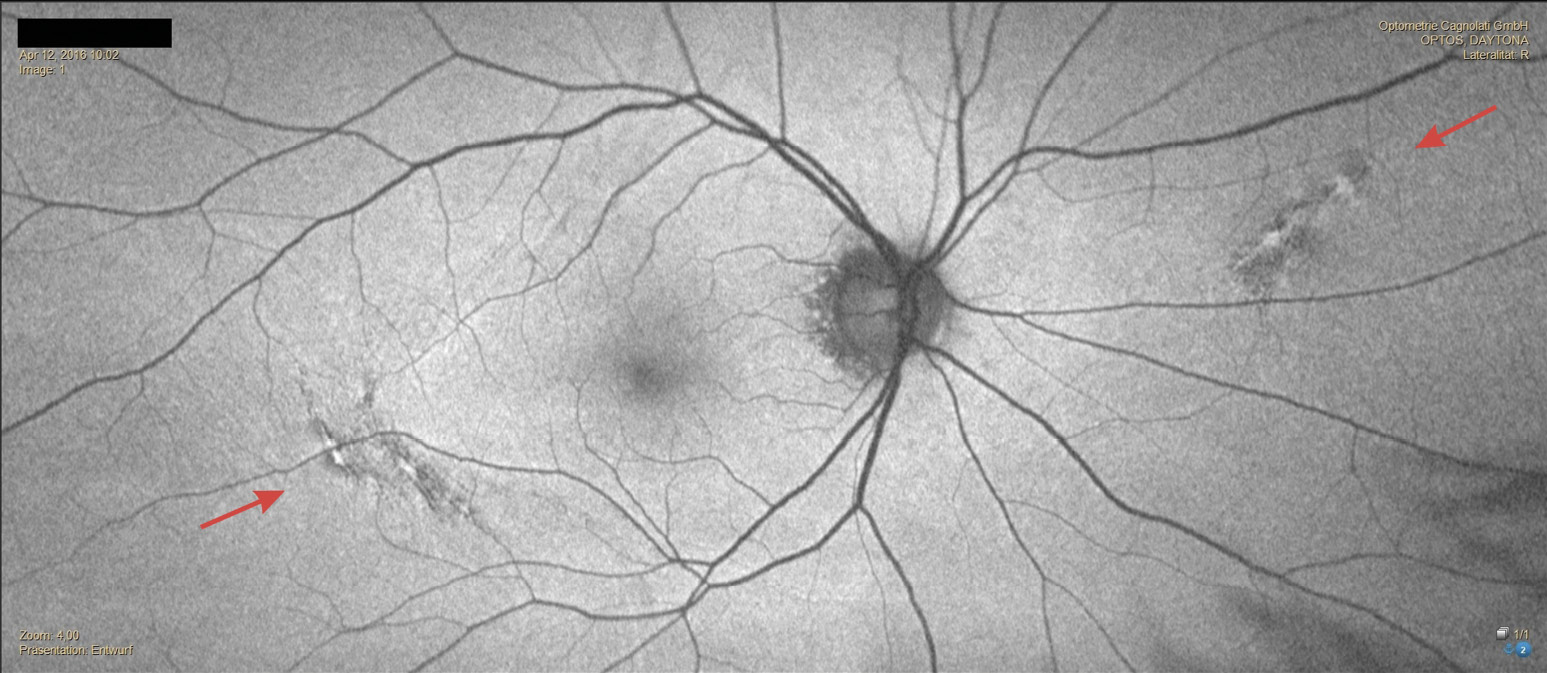
In April 2016, the patient returned for a comprehensive eye examination. Except for reduced near vision quality, he remained asymptomatic. The refraction for distance vision showed no changes in power. Only the power of the near addition had to be increased by 0.5 D due to a slight reduction in the amplitude of accommodation. The corrected distance visual acuity was unchanged at OD/OS: 20/25 and OS: 20/20 p. The examination of the anterior segment of the eye corresponded to the results from 2013. The IOP was OD: 18 mmHg and OS: 19 mmHg (11:00 a.m.). The retinal pigment changes had increased in both eyes. They looked like a network of pigmented lines and nodules. (Figure 3 and Figure 4). Due to the significant increase in the pigmentary changes described in the fundus, we now referred the patient for further evaluation by an ophthalmologist.
Examination in December 2024
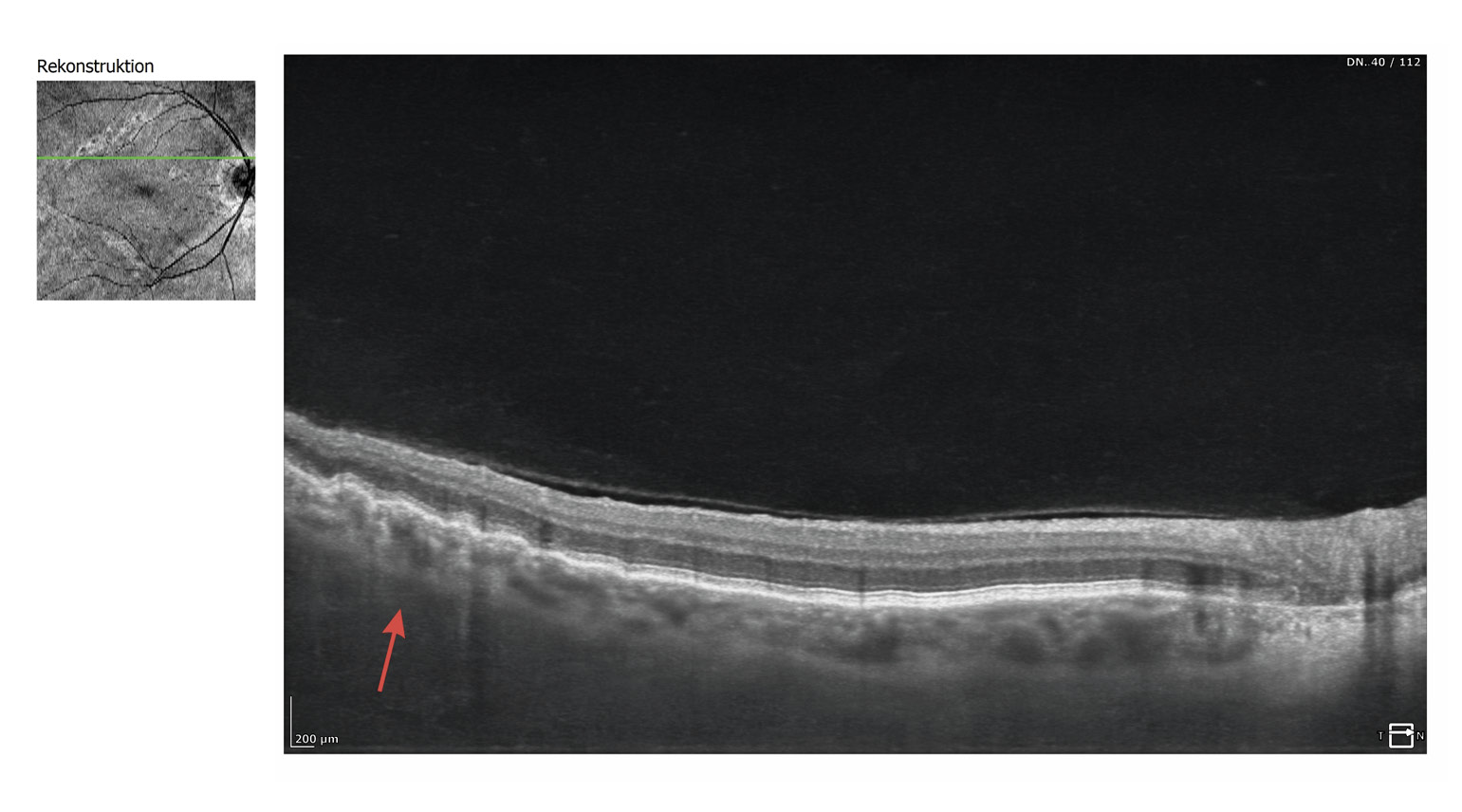
In December 2024, the patient consulted us again and complained of reduced near vision quality. He also informed us that he had last visited an ophthalmologist in 2021 and that the ophthalmologist had diagnosed mild AMD based on the pigmentary changes described in the fundus, but that this was not a reason for concern due to his good visual acuity. The refraction we performed for distance vision showed significant changes in power. The newly determined refraction and visual acuity values were as follows: OD: +2.50 D cyl −2.75 D axis 180° and OS: +2.25 D cyl −2.75 D axis 1° with a monocular visual acuity of OD: 20/25 and OS: 20/20 and a binocular visual acuity of 20/20. The refractive power of the near addition had to be increased to 2.5 D due to the slightly reduced accommodation amplitude. The examination of the anterior segment of the eye corresponded to the results from 2016; only the crystalline lens in both eyes showed a beginning cataract. The IOP was OD: 19 mmHg and OS: 19 mmHg (12:00 o‘clock). In the posterior segment of the eye, there was a significant increase in pigmentary changes in both eyes, both in the area of the optic disc and macula and in the central fundus. The lesions were noticeably enlarged in both eyes. Despite the more pronounced lesions in the right eye, symmetry of the lesions between both eyes was evident. The hypo- and hyperfluorescent areas formed a distinct network of pigmented lines and nodules resembling a fishing net (Figures 5, 6, 7, 8, 9) which is why, after consulting another clinician, a reticular dystrophy of the retinal pigment epithelium could be assumed.
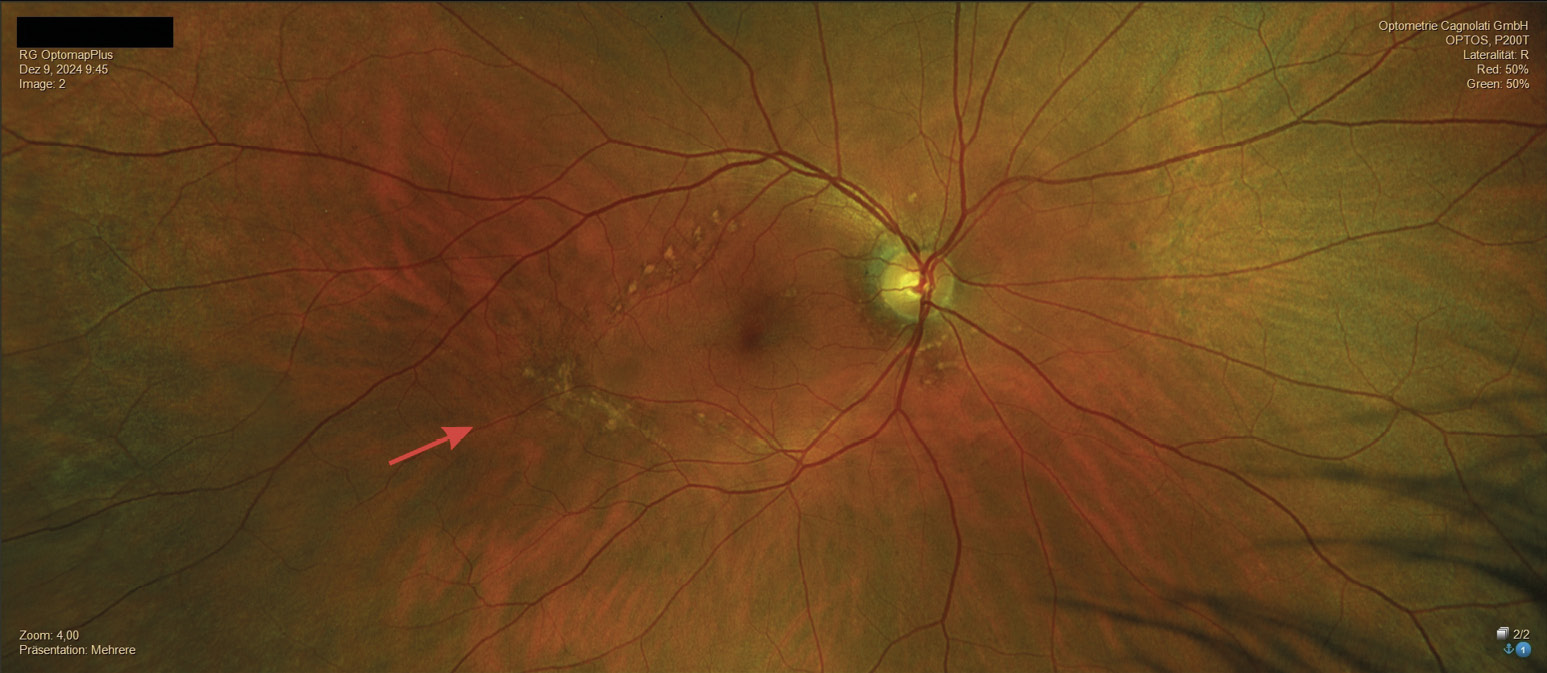
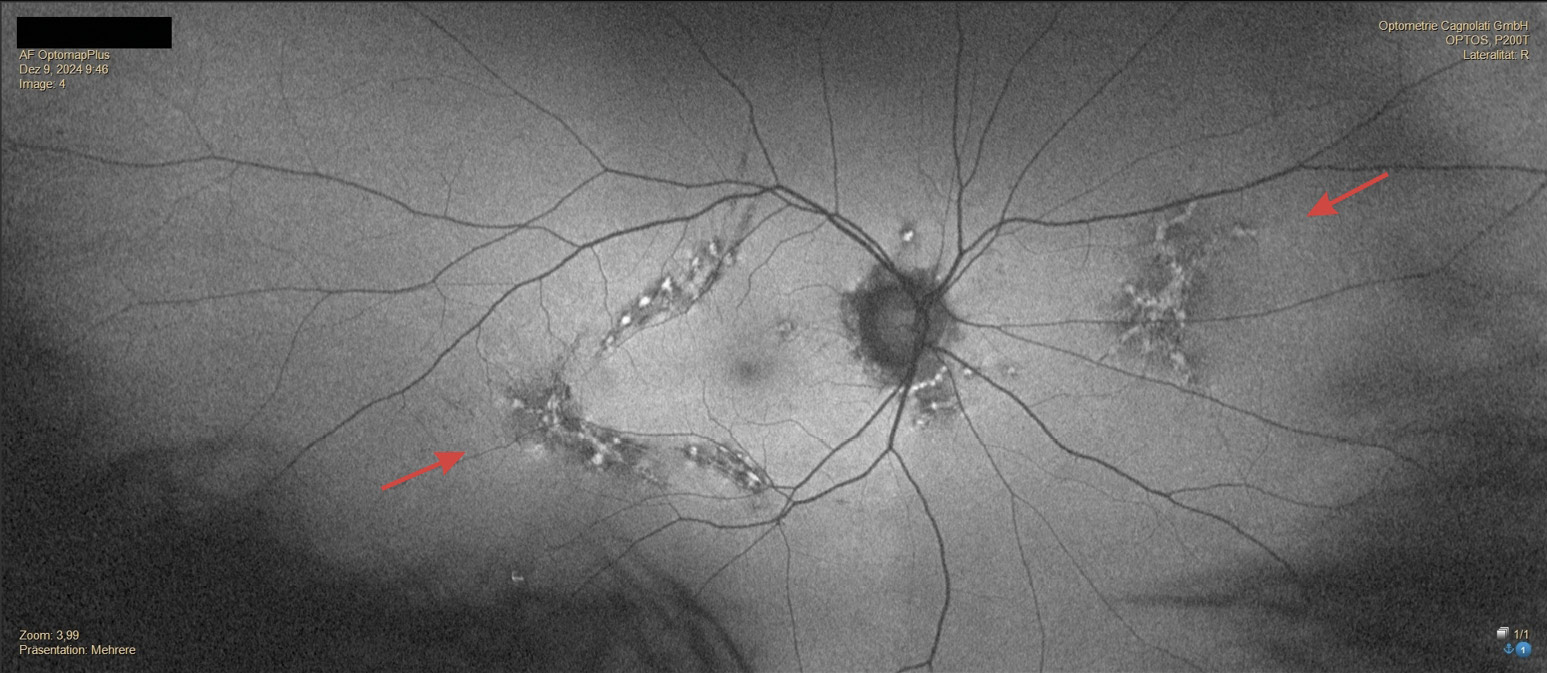
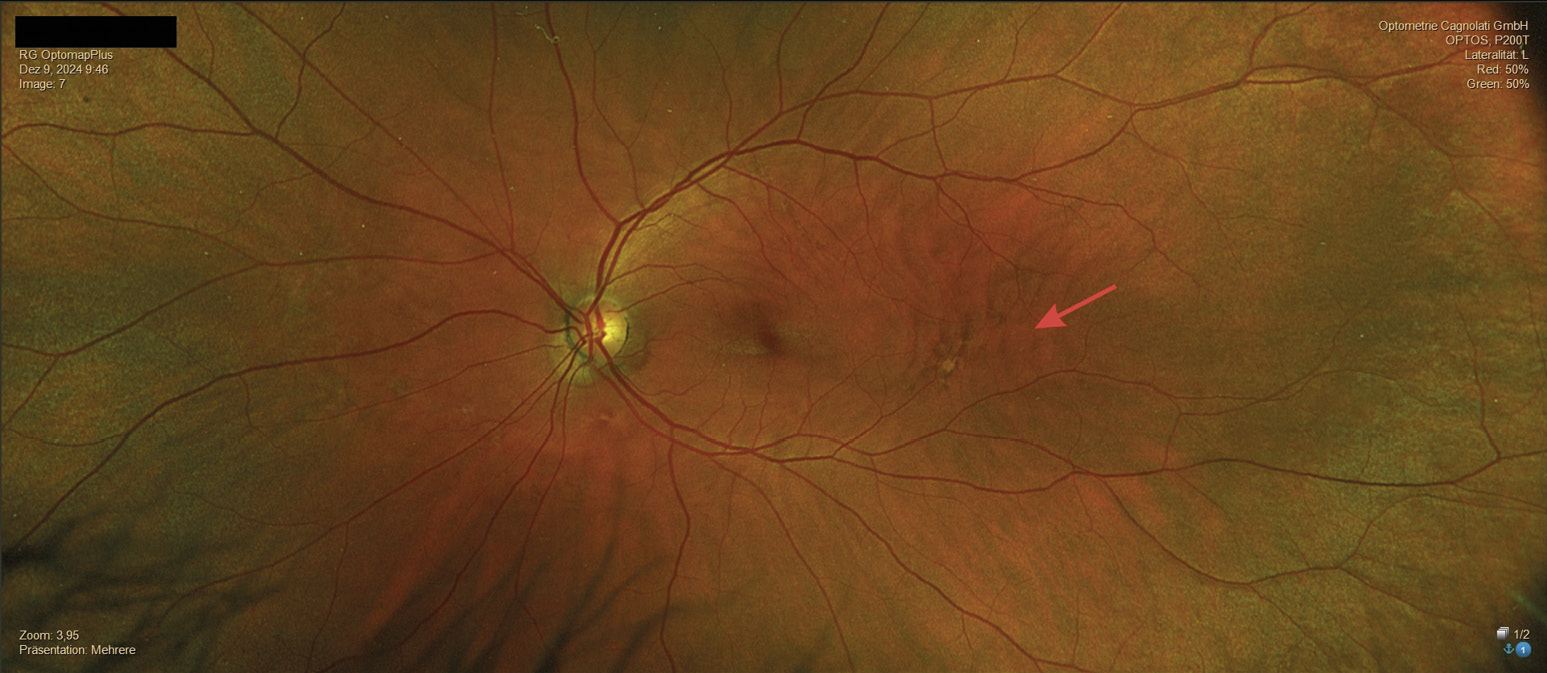
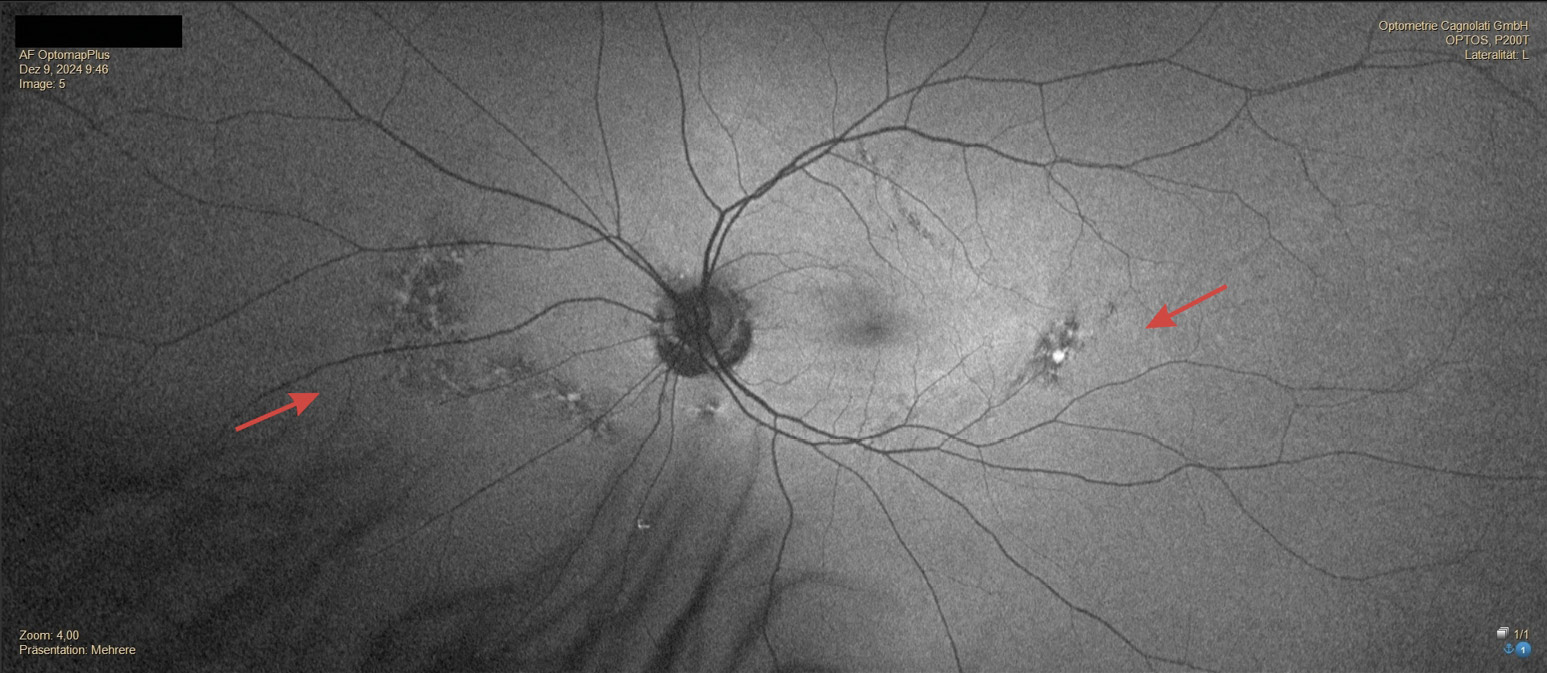
Discussion
Reticular dystrophies of the retinal pigment epithelium (RPE) can start in childhood.10 Patients with pattern dystrophy of the RPE usually remain asymptomatic until their fourth or fifth decade of life.2 Symptoms are often only noticed due to deterioration in central vision. Pattern dystrophies are frequently confused with AMD, although the loss of visual acuity in patients with pattern dystrophy is usually significantly less than in AMD patients.7 Zerbib et al. reported that the lesions in reticular dystrophy differ from those in other macular dystrophies. However, they are more similar to the characteristics of fundus flavimaculatus than to those of foveomacular vitelliform dystrophy in adults.11 Chen et al. determined that the function of the blood-aqueous barrier may be altered in reticular dystrophy of the retinal pigment epithelium.12 There are still relatively few case reports on reticular dystrophy in the literature. In a publication from 1976, British ophthalmologist Chopdar cites a total of three publications on this topic.13 In their 2023 publication, Modrzejewska et al. refer to a total of ten clinical publications worldwide on this topic.6 As with all pattern dystrophies, reticular dystrophy also involves local lipofuscin accumulation in the RPE. Lipofuscin is located above the RPE and below the photoreceptors in the interdigitation zone of the retina.7 Fluorescein angiography (FA) or fundus autofluorescence (FAF) shows areas of hypofluorescence surrounded by areas of hyperfluorescence and normal choroidal vascular structure.14 Reticular dystrophy is usually detected incidentally during an eye examination. In addition to ophthalmoscopic examination of the retina, retinal imaging using OCT and autofluorescence is helpful for diagnosis. Electroretinography (ERG) and electrooculography (EOG) are usually normal or minimally abnormal in reticular dystrophy.2
Conclusion
Pattern dystrophies of the retinal pigment epithelium (RPE) are still considered relatively rare findings today, although their incidence appears to be significantly higher. Since most patients are usually asymptomatic until their fourth or fifth decade of life, pattern dystrophies are often only discovered as incidental findings during routine eye examinations. They are often confused with AMD. Although the visual prognosis for affected patients is good, patients should be monitored regularly for possible atrophy and/or choroidal neovascularization. The current case report provides additional information on the long-term progression of reticular dystrophy of the retinal pigment epithelium over a period of 11 years.
Acknowledgements
The author would like to thank Dr. Sherry Bass, from the State University New York (SUNY), College of Optometry, for her valuable contributions during the preparation of the manuscript.
Conflict of interest
The author has given seminars and workshops for Optos GmbH in the past and has received a honorarium for these activities.