West African Crystalline Maculopathy: A Case Report
Purpose: Crystalline maculopathies can have numerous etiologies with one of the less common etiologies being West African crystalline maculopathy. This case report reviews the case of a patient found to have West African crystalline maculopathy, the cause of this maculopathy, and its differentials.
Material and Methods: A 79-year-old African American female with a medical history significant for diabetes mellitus and hypertension was examined for a routine diabetic eye exam. It was found that she had yellow crystalline-like deposits in the macular region of her right and left eye. Fundus images were obtained along with optical coherence tomography (OCT) images.
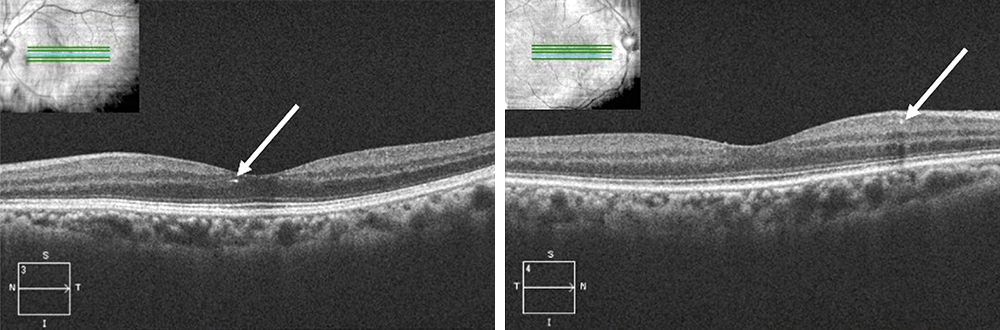
Results: Dilated fundoscopy showed that the patient had multiple yellow crystalline deposits in the macular region of both eyes. OCT imaging showed that these deposits were localized to the inner retinal layers. The patient reported that she was from the Igbo tribe, located in southeast Nigeria. Additional history included a diet high in kola nuts.
Conclusion: West African crystalline maculopathy is a form of crystalline maculopathy that was initially discovered in patients from the Igbo tribe from southeast Nigeria. While the origin of these crystalline deposits is not clear, it is suspected that high amounts of kola nuts in the diet is a significant risk factor in the development of this form of maculopathy.
Introduction
A 79-year-old African American female presented for a routine diabetic eye exam. Her only complaint was of mild irritation, greater in the left eye than the right eye, which was relieved with use of artificial tears. Her past ocular history was significant for grade 1 hypertensive retinopathy, age-related cataracts, and dry eye syndrome in both eyes. She was noted to have epiretinal membrane without traction in both eyes and exudative material in the macula as per previous records. Her medical history was significant for type II diabetes mellitus for 13 years. Her last hemoglobin A1c level was 7.1% and was controlled with oral medications, glimepiride and linagliptin. Her last glucose levels were last measured to be 188mg/dL with blood work. Her medical history was also remarkable for hypertension, which was controlled with amlodipine-benazepril, and hyperlipidemia, which was controlled with simvastatin.
Material and Methods
The patient was examined with a dilated fundus exam at a New York City Health + Hospitals Corporation hospital in New York State, USA. Fundus images were obtained with the Zeiss Visucam Pro retinal camera (Zeiss International, Oberkochen, Germany) and optical coherence tomography (OCT) was performed with the Zeiss Cirrus 4000 (Zeiss International, Oberkochen, Germany).
Results
Best corrected visual acuity at distance was found to be 20/20 (logMAR 0.00) in the right eye and 20/25 (logMAR 0.10) in the left eye with a refraction of +3.50-1.00x085 and +3.25-1.00x090 in the right and left eye respectively. Near visual acuity was also found to be 20/20 (logMAR 0.00) and 20/25 (logMAR 0.10) in the right and left eye with an add power of +2.50. Anterior segment evaluation was significant in both eyes for capped Meibomian glands, frothy tear film at the lid margins, and a reduced tear break-up time. Dilated examination demonstrated nuclear changes with anterior and posterior cortical changes to the crystalline lens in the right and left eye with the left eye also showing evidence of central vacuoles through the visual axis. The patient was noted to have a posterior vitreous detachment in the right eye only.
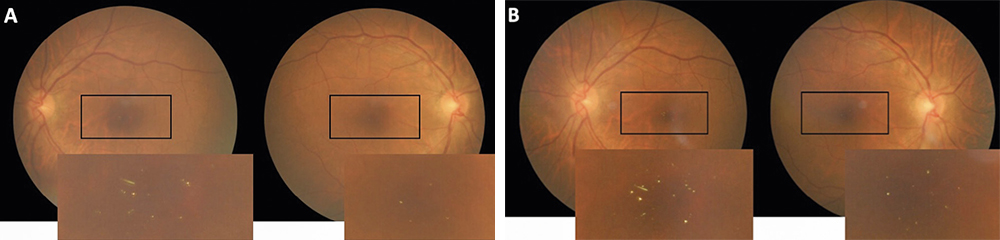
Fundoscopy showed evidence of attenuated retinal arterioles in both the right and left eye. There was no evidence of hemorrhaging, vascular changes, or neovascularization consistent with diabetic retinopathy in either eye. Macular examination demonstrated the appearance of multiple small yellow hyper-reflective deposits within the superficial retina that was greater in the left eye than the right eye. Fundus photos were taken to document these hyper-reflective deposits (Figure 1) and OCT scans were performed. The OCT scans through the hyper-reflective deposits demonstrated that these deposits were located within the inner retinal layers, from the inner nuclear layer to the ganglion cell layer (Figure 2). Upon further questioning, the patient reported that she is from the Igbo tribe in southeast Nigeria. Her dietary consumption was significant for kola nuts. She denied any history of intravenous drug use, prior use of tanning agents, and had no reported history of breast cancer or tamoxifen use. Fundus photos from the current exam (Figure 1A) can be compared to the photos from 3 years prior (Figure 1B).
Discussion
West African crystalline maculopathy is a form of maculopathy first described by Sarraf et al.1 in 2003 when a cluster of six patients in Los Angeles, California was found to have crystalline maculopathy. All six of the patients were discovered to have originated from the Igbo tribe in southeast Nigeria. At this time, the prevalence rate of this condition has not been determined. This maculopathy presents as multiple small refractile crystals in the macular and paramacular region that are typically asymmetric They appear to have no effect on visual acuity and require no intervention. The patient discussed above showed no adverse visual effects from the crystalline maculopathy and is currently being monitored annually with serial imaging. While the etiology and composition of these crystals is unclear, it has been proposed that the ingestion of kola nuts may be associated with the prevalence of this type of maculopathy.1 The kola nut is an important part of the diet in West Africa and is seen as a powerful cultural symbol in the region.2 While the effects of kola nuts on the human body have not been thoroughly studied, it is believed that it can be used as a remedy for a diverse number of maladies including but not limited to fatigue, hunger, depression, irregular menstrual cycles, and morning sickness.2-3 A study of students in Osogbo, Nigeria found that the lifetime usage rate of kola nut chewing was 74.8%, with the majority of users starting at age 14 or below. Increased usage was associated with poorer school attendance and lower age group.4 A major component of the kola nut is caffeine, which can have a stimulant effect, contributing to increased alertness.5
It is unclear if the crystals are due to a component of the kola nut as its consumption is widespread throughout Nigeria while the prevalence of West African crystalline maculopathy is not consistent with that widespread consumption. However, there are many foods and spices that are found more commonly in the West African diet6 suggesting there may be other dietary considerations besides kola nuts resulting in crystalline maculopathy. There is evidence to suggest that retinal vascular disease may be a risk factor in this crystalline maculopathy. Oluleye et al. reported two cases of a crystalline maculopathy in females from the Yoruba tribe of western Nigeria that reported no history of kola nut consumption.7 However, these patients’ medical history was significant for diabetes mellitus.
Two case series by Browning and Morettin et al. described patients who presented with crystalline deposits in the macula that also had a history of diabetes mellitus.8-9 Of the three patients described by Browning, two were of Nigerian origin (from Ibibio tribe and Igbo tribe) and one was of Liberian descent (from the Bassa tribe) and only one had a history of kola nut consumption. All three had a history of diabetes mellitus and some degree of diabetic retinopathy. It has been hypothesized that diabetic retinopathy is a predisposing factor for crystalline maculopathies.8 Browning also noted that the pattern of crystals may change, with possible resorption of some crystals, and postulated it to be a result of laser photocoagulation treatment for diabetic retinopathy. The patient described above also showed signs of potential resorption of crystals, however she had no history of focal laser treatment. In our patient, it was difficult to ascertain if the fewer number of crystals seen in the current photographs were due to resorption of the crystals over time or the slight reduction in image quality from cataract progression.
Of the two patients described by Morettin et al., one patient emigrated from Tema, Ghana and the other from Nigeria. One of the patients had a medical history significant for diabetes mellitus with diabetic retinopathy and reported no history of kola nut consumption. The second patient did report kola nut consumption but had no history of diabetes, however she did have a vascular risk factor in hypertension with hypertensive retinopathy.9 Kung et al. reported a case of a patient from the Benin tribe of Nigeria that presented with signs consistent with West African crystalline maculopathy with concurrent diabetes mellitus type II, hypertension, and sickle cell.10 There was no reported consumption of kola nuts. The patient presented with unilateral macular edema consistent with diabetes and was also found to have crystalline deposits in both eyes. However, the crystalline deposits were found to be greater in the eye with the macular edema suggesting there may be a correlation between the level of retinopathy and presence of West African crystalline maculopathy.
A review of 14 patients that presented with West African crystalline maculopathy by Rajak et al. supports this correlation of crystalline deposits and level of diabetic retinopathy.11 These 14 patients originated from different West African countries including Nigeria, Sierra Leone, and Ghana. Five patients denied any history of kola nut consumption, however all 14 patients presented with coexistent retinal vascular pathologies. These pathologies included diabetic maculopathy, sickle cell retinopathy, branch macular vein occlusion, and familial exudative vitreoretinopathy. One patient presented with unilateral macular crystals in an eye that had a branch retinal vein occlusion. It was proposed that these crystals form after there is a breakdown of the blood-retina barrier in patients with preexisting retinal vascular disease.11 These cases suggest that retinal vascular disease may be a significant risk factor in the pathogenesis of these crystalline deposits.
As there are several different etiologies for crystalline maculopathy12, other treatable forms of crystalline maculopathy should be ruled out. These maculopathies can be ruled out based on the patient’s history, use of certain medications and a diabetic work-up. The following are some differentials that should be considered in a patient with crystalline maculopathy:
• Medication
- Tamoxifen is a nonsteroidal antiestrogen and immune modulator13 that is used in the treatment of estrogen positive breast cancer. Retinal side effects of tamoxifen are described as white refractive retinal opacities, most commonly located in the outer plexiform layer.14
- Methoxyflurane is an analgesic used for pain management and anesthesia.15 Prolonged exposure can result in calcium oxalate crystals in the retinal pigment epithelium, neurosensory retina, and other areas of the body.16
- Nitrofurantoin is a synthetic antibiotic used in the treatment of urinary tract infection. Long-term treatment can result in superficial and deep intraretinal glistening deposits in a circinate pattern.17
- Flupentixol is an antipsychotic neuroleptic drug used in the treatment of schizophrenia. There is a weak correlation between long-term use of flupentixol and yellow-white refractile intraretinal deposits in the macular and peripapillary area.18
• Canthaxanthin is a carotenoid pigment with a red to orange hue that is used in food dye and tanning pills. Total consumption of high doses of canthaxanthin can result in reflective crystals that accumulate in the perifoveal area, located in the outer plexiform layer.19 There may be reduced visual acuity and visual field defects, although most patients are asymptomatic.
• Exudates found in the macular and paramacular region from diabetic retinopathy can result in the appearance of yellow glistening material that may resemble refractile crystals.
• Talc is a filler substance often used in narcotics such as heroin. Small particles of talc can enter the retinal vessels and result in microemboli in vessel walls.20 These small deposits can mimic the appearance of crystals, although they are located within the retinal vasculature rather than the intraretinal layers.
Conclusion
West African crystalline maculopathy is an uncommon crystalline maculopathy found within tribes in West African nations. While the etiology of these crystals is unknown, there is evidence that a combination of West African specific diet and retinal vascular disease can contribute to the formation. Since these crystals are not visually significant, no intervention is required, and can be monitored with serial photography.
Conflict of interest
The author declares that there is no conflict of interests regarding the methods and devices mentioned in the article.
